This chapter aims to inform policy makers of the choice of policies to tackle antimicrobial resistance (AMR) in the human health, animal and environmental sectors. For each sector, the chapter starts by describing policy options aiming to address the main drivers of AMR. It then presents an overview of the current state of evidence, with a particular focus on the design features and contextual factors that can enable or hinder the effectiveness of each policy. Throughout, a number of global AMR initiatives and good practice examples are documented. The chapter concludes by providing a summary of key lessons emerging from the literature.
Embracing a One Health Framework to Fight Antimicrobial Resistance

5. Tackling antimicrobial resistance in One Health framework: Policy approaches
Abstract
Key messages
Since the previous OECD publication Stemming the Superbug Tide: Just a Few Dollars More (2018[1]), evidence based on AMR interventions in the human health sector has grown. Complementing previous OECD work, the current analysis shows that, in human health, prudent use of antibiotics can be promoted through: an array of pharmaceutical regulations; interventions that support healthcare providers’ prescribing decisions at the point of care; and information-based strategies focusing on improving the general public’s knowledge and perceptions around antimicrobials.
The introduction and scale‑up of a wide range of infection prevention and control (IPC) interventions have been shown to reduce the burden of resistant healthcare‑associated infections (HAIs) and strengthen AMR surveillance. Specifically, the current analysis looks at the available evidence that demonstrates the effectiveness of selected IPC interventions on curbing AMR, including: integrating AMR in HAI surveillance; IPC monitoring, evaluation and feedback interventions; creating dedicated IPC leadership at health facilities that promote IPC training and education activities and encourage greater compliance with IPC guidelines; and optimising the organisation of care. Available evidence also suggests that the effectiveness of measures to promote prudent use of antimicrobials may be enhanced if implemented in conjunction with IPC interventions.
Public health programmes to improve vaccination coverage can help interrupt AMR transmission. Broadly, vaccination coverage among OECD members remains relatively high but some countries are experiencing significant challenges in maintaining performance partly due to hesitancy towards the available vaccines. A range of individual- and community-level interventions have been suggested as effective tools to curb vaccine hesitancy across OECD countries.
Globally, food consumption is rising faster than production, exerting pressure on producers to resort to intensive agricultural practices in livestock and crop production that depend on antimicrobials. Regulations that promote the prudent use of veterinary antimicrobials, particularly those that limit the use of antimicrobials as growth promoters, have been shown to reduce the AMR burden.
In addition, enhancements in farm management and biosecurity practices and improvements in the coverage of animal vaccines have been shown to reduce the likelihood of resistant infections emerging and spreading among animal and human populations. In plant production, there are important cross-country gaps in regulatory frameworks relevant to the use of antimicrobials, as well as notable challenges in the existing capacity to monitor and evaluate the changes in antimicrobial use over time.
Supplementing efforts to improve animal and plant health, the rollout and implementation of rigorous food safety compliance systems have been associated with reductions in AMR transmission across the food supply chain.
Emerging evidence suggests that improvements in wastewater treatment facilities as well as waste management practices in pharmaceutical manufacturing, agricultural production and healthcare settings are linked with reductions in the transmission of AMR by promoting the safe disposal and removal of antimicrobials from the environment. But available evidence suggests that these measures alone are unlikely to halt AMR transmission in the environment due to the limitations of the existing technologies.
In addition, scaling up drug take‑back programmes for households offers a promising avenue for interrupting AMR transmission in the environment.
Key lessons emerging from the literature
In human and animal health, flexible AMR policies that aim to create an enabling work environment that promotes prudent use of antimicrobials achieve similar levels of improvements in antibiotic prescribing behaviours compared to restrictive policies that limit the opportunities for using antibiotics.
Getting input and buy-in from key stakeholders (e.g. healthcare providers, veterinarians, farmers) in the design and implementation of AMR policies improves the effectiveness of these policies.
The effectiveness of AMR policies may change over time, necessitating modifications in the design and implementation of these policies to address evolving needs in a given setting.
Even when AMR interventions are effective, unintended consequences may occur.
Improving the availability and accessibility of information from novel data sources offers an important avenue for supporting rigorous evaluations of AMR policies in line with the One Health framework.
Since the last OECD publication, the evidence base on the effectiveness of AMR-relevant policies has grown
In 2018, the OECD published a landmark report – Stemming the Superbug Tide: Just A Few Dollars More – which evaluated the health and economic impact of AMR (OECD, 2018[1]). Using the OECD Strategic Public Health Planning for AMR model and advanced modelling techniques, this publication showed that AMR rates are high and estimated to grow in Group of Seven (G7) countries, OECD members and key partners. A key contribution of this publication was a review of policies to tackle the growing AMR burden in the human health sector. Since this publication, the evidence base that sheds light on the effectiveness of AMR policies in human health and other sectors has grown. The complexities around designing and implementing policies to tackle AMR means it is imperative to re‑examine the emerging evidence on the effectiveness of various policy options in line with the One Health framework (see Chapter 4). In the context of the ongoing COVID‑19 pandemic, concerted efforts to tackle AMR remain ever more pertinent, which emphasise the importance of the complex interlinkages between human, animal and environmental health (Box 5.1).
Box 5.1. A threat or an opportunity: What does the COVID‑19 pandemic mean for tackling AMR?
The ongoing pandemic may present a threat or an opportunity for efforts to stem the AMR tide. The COVID‑19 pandemic threatens to accelerate the burden of AMR in several ways:
In many countries, antibiotic stewardship programmes (ASPs) are experiencing disruptions as health workers are diverting efforts away from core ASP activities to pandemic response (see Chapter 4).
Adherence to the World Health Organization (WHO) guidance on antibiotic use for COVID‑19 patients appears low. The WHO recommends antibiotic treatment for mild/moderate COVID‑19 cases only if signs of a bacterial infection are present (Getahun et al., 2020[2]). Yet, two recent literature reviews reported that antibiotics were used to treat 72‑74.6% of hospitalised COVID‑19 patients (Rawson et al., 2020[3]; Langford et al., 2021[4]), while it is estimated that only about 8% of COVID‑19 patients reported having bacterial and fungal co‑infections (2020[3]). Similarly, Langford et al. (2021[4]) suggested that bacterial co‑infections among COVID‑19 patients were around 8.6%.
Increases in hospitalisations may elevate the risk of healthcare‑associated infections (HAIs) and transmission of multidrug-resistant organisms (Saleem et al., 2019[5]). This elevated risk may be exacerbated by increased workload among health workers and fluctuations in compliance with infection prevention and control (IPC) measures in healthcare settings. This was evident during the 2003 SARS epidemic, with evidence suggesting that poor compliance with IPC measures was linked to increases in the acquisition of methicillin-resistant Staphylococcus aureus (MRSA) (Yap et al., 2004[6]).
Service delivery disruptions for non-COVID patients can hinder efforts to curb AMR. To date, several systematic reviews looked at disruptions in service delivery based on data gathered primarily from OECD countries. One review concluded that, among patients who suffer from acute cardiovascular disease, the COVID‑19 pandemic resulted in significant reductions in the rate of hospital admissions and the number of medical procedures carried out (Kiss et al., 2020[7]). This study also showed that the length of hospital stay for these patients was shortened, while there were longer delays between the onset of symptoms and treatment at a hospital. Another systematic review examined evidence from France, Italy, South Korea and the United States (US) and concluded that there were notable delays in the provision of emergency medical services outside hospital settings for patients suffering from cardiac arrest (Scquizzato et al., 2020[8]). Similar delays and disruptions were observed in routine cancer care (Riera et al., 2021[9]).
The ongoing pandemic also offers important lessons:
Building on lessons learnt during the COVID‑19 pandemic is paramount to tailoring ASPs for future health emergencies. Moving forward, ASPs can be updated to cover the rapid changes in healthcare delivery modalities to respond to evolving health needs. For instance, to date, integrating relatively newer modes of care (e.g. telemedicine) into ASPs was shown to yield beneficial impacts in promoting the prudent use of antibiotics and reducing AMR (dos Santos et al., 2018[10]).
The COVID‑19 pandemic brought renewed attention to the importance of comprehensive IPC strategies to curtail HAIs in the context of health emergencies (Lucien et al., 2021[11]). One living systematic review by Chou et al. (2020[12]) shows that, in healthcare settings, IPC measures were linked with reductions in the probability of infections among health workers in the context of the current and previous outbreaks like SARS-CoV‑1 and MERS-CoV. Specifically, global evidence is mounting on the effectiveness of enhancing hand hygiene practices, IPC training and education and personal protective equipment (Chou et al., 2020[12]). Lessons learnt from countries that have explicitly integrated IPC strategies in the context of the ongoing pandemic can foster further improvements in IPC practices.
The rapid development of COVID‑19 vaccines demonstrated that medical innovations can be fast-tracked. Prior to COVID‑19, the fastest vaccine development took place in the 1960s for tacking mumps (Bloom et al., 2021[13]). In many countries, COVID‑19 vaccination programmes are being rolled out at an unprecedented rate, providing lessons for tackling demand- and supply-side barriers that may impede the accessibility and use of vaccines.
Innovative modes of inter-governmental co‑operation and financing platforms emerged since the outset of the pandemic. These experiences proved crucial for scaling up vaccine research efforts in multiple settings at the same time (Bloom et al., 2021[13]). Lessons learnt from these experiences can provide a powerful means for scaling up research and development (R&D) initiatives in support of the development of new antimicrobials.
Source: Getahun, H. et al. (2020[2]), “Tackling antimicrobial resistance in the COVID‑19 pandemic”, https://doi.org/10.2471/blt.20.268573; Rawson, T. et al. (2020[3]), “Bacterial and fungal co‑infection in individuals with coronavirus: A rapid review to support COVID‑19 antimicrobial prescribing”, https://doi.org/10.1093/cid; Langford, B. et al. (2021[4]), “Antibiotic prescribing in patients with COVID‑19: Rapid review and meta‑analysis”, https://doi.org/10.1016/j.cmi.2020.12.018; Saleem, Z. et al. (2019[5]), “Point prevalence surveys of health-care‑associated infections: A systematic review”, https://doi.org/10.1080/20477724.2019.1632070; Yap, F. et al. (2004[6]), “Increase in methicillin-resistant Staphylococcus aureus acquisition rate and change in pathogen pattern associated with an outbreak of severe acute respiratory syndrome”, https://doi.org/10.1086/422641; Kiss, P. et al. (2020[7]), “The impact of the COVID‑19 pandemic on the care and management of patients with acute cardiovascular disease: A systematic review”, https://doi.org/10.1093/ehjqcco/qcaa084; Scquizzato, T. et al. (2020[8]), “Effects of COVID‑19 pandemic on out-of-hospital cardiac arrests: A systematic review”, https://doi.org/10.1016/j.resuscitation.2020.10.020; Riera, R. et al. (2021[9]), “Delays and disruptions in cancer health care due to COVID‑19 pandemic: Systematic review”, https://doi.org/10.1200/go.20.00639; dos Santos, R. et al. (2018[10]), “Antimicrobial stewardship through telemedicine and its impact on multi-drug resistance”, https://doi.org/10.1177/1357633x18767702; Lucien, M. et al. (2021[11]), “Antibiotics and antimicrobial resistance in the COVID‑19 era: Perspective from resource‑limited settings”, https://doi.org/10.1016/j.ijid.2020.12.087; Chou, R. et al. (2020[12]), “Epidemiology of and risk factors for coronavirus infection in health care workers”, https://doi.org/10.7326/m20-1632; Bloom, D. et al. (2021[13]), “Moving beyond traditional valuation of vaccination: Needs and opportunities”, https://doi.org/10.1016/j.vaccine.2016.12.001.
The goal of this chapter is to review the global evidence on policy options to tackle AMR building on previous OECD analysis. This chapter complements the first policy chapter, which presents the global progress in the implementation of AMR policy priorities highlighted in the WHO Global Action Plan on Antimicrobial Resistance (GAP AMR) and the content of action plans from selected OECD, European Union/European Economic Area (EU/EEA) countries and Group of Twenty (G20) countries. Although the focus of this chapter is on human health (Box 5.2), policies concerning animal health, plant health, agri-food systems and the environment are also presented in line with the One Health approach. To be as useful as possible for policy makers, each section is organised according to the aim of each policy option in different thematic areas. The choice of evidence presented in this chapter is purposeful. While the chapter aims to be as comprehensive as possible in its synthesis of evidence, it does not attempt to present a summary of all available evidence on all policy options. Due to data limitations, most evidence comes from the OECD, EU/EEA and G20 countries, as well as OECD partners. Studies from low- and middle‑income countries (LMICs) are presented whenever possible.
Box 5.2. AMR-relevant policies in human health covered in previous and current OECD analyses
The previous and current OECD analyses complement each other (Figure 5.1). The previous OECD publication – Stemming the Superbug Tide: Just a Few Dollars More – focused on a wide array of policies in human health (2018[1]). Since then, the evidence base on the effectiveness of these interventions has grown, further underscoring the importance of each strategy. This chapter expands the scope of previous OECD analyses. Whenever possible, the chapter emphases the design features of AMR policies and contextual factors that can enable or hinder the effectiveness of AMR policies. In line with the One Health framework, the chapter also presents lessons emerging from different strands of the literature.
Figure 5.1. AMR-relevant policies included in previous and current OECD analyses
Source: OECD (2018[1]), Stemming the Superbug Tide: Just a Few Dollars More, https://doi.org/10.1787/2074319x.
Policies to tackle AMR in human health
Policies to promote prudent use of antibiotics
Antibiotic stewardship programmes (ASPs)
Policy interventions
Persuasive interventions aiming to create an enabling environment for prudent use of antibiotics.
Restrictive strategies limiting opportunities to use antibiotics.
Strategies targeting structural elements of care.
Key messages
ASPs are effective in reducing imprudent use of antibiotics without increasing the risk of death.
Restrictive or persuasive ASPs can be effective in reducing imprudent use of antibiotics. Supplementing restrictive interventions with persuasive ones enhances the effectiveness of the former.
Effectiveness of ASPs will be enhanced by tracking performance over time in accordance with the context of care.
Effectiveness of ASPs can be elevated by addressing the existing gaps in the available antibiotic guidance and extending guidance for relatively new modes of healthcare delivery such as telehealth.
In countries where the prevalence of informal healthcare providers is high, addressing antibiotic prescription outside of clinical settings is crucial to support efforts to build effective ASPs in clinical settings.
Since the release of Stemming the Superbug Tide: Just a Few Dollars More (OECD, 2018[1]), empirical evidence on the effectiveness of ASPs in different healthcare settings has accumulated. ASPs have been shown to effectively reduce imprudent use of antibiotics without exacerbating the risk of mortality (Davey et al., 2017[14]). In hospital settings, ASPs have been linked to reductions in the duration of antibiotic treatment, shorter hospital stays (Van Dijck, Vlieghe and Cox, 2018[15]; Honda et al., 2017[16]; Nathwani et al., 2019[17]) and lower treatment costs, though the degree to which countries realise savings in costs varies across settings (Honda et al., 2017[16]; Nathwani et al., 2019[17]). While the expansion of the analytical base on the effectiveness of ASPs is encouraging, further improvements are needed in methods used to assess the impact of ASPs (Schweitzer et al., 2019[18]; de Kraker et al., 2017[19]).
In 2019, the WHO published a practical toolkit that provides guidance for ASPs in healthcare settings, which groups ASPs into three broad categories as shown in Table 5.1 (WHO, 2019[20]):
Persuasive strategies that rest on provider education and feedback efforts to induce behaviour change.
Restrictive strategies that limit opportunities to use antibiotics.
Structural strategies that target organisational elements of care.
Table 5.1. WHO groupings of AMR interventions to improve antibiotic prescribing behaviours in healthcare settings
|
Intervention type |
Example interventions |
|---|---|
|
Persuasive (education) |
|
|
Persuasive (feedback) |
|
|
Restrictive |
|
|
Structural |
|
Source: Adapted from WHO (2019[20]), Antimicrobial Stewardship Programmes in Health-care Facilities in Low- and Middle‑income Countries: A WHO Practical Toolkit, https://apps.who.int/iris/handle/10665/329404.
The design and implementation of ASPs vary substantially across countries but useful lessons emerge. The WHO guidance indicates that restrictive interventions can yield relatively quick gains in antibiotic use but the effectiveness of these interventions reaches similar levels compared to those achieved through persuasive interventions around a six‑month time frame (WHO, 2019[20]). In congruence with the WHO guidance, one recent systematic review suggested that both restrictive and persuasive policies can achieve improvements in antibiotic behaviours at similar magnitudes and that supplementing restrictive interventions with persuasive ones may augment the effectiveness of the former (Davey et al., 2017[14]). Emerging evidence also points to promising results in improvements in antibiotic behaviours among providers in response to structural strategies (WHO, 2019[20]).
The effectiveness of ASPs can be improved by embedding measurement frameworks that track performance over time. Yet, an important limitation of many ASPs is that they set out ambitious targets for promoting the prudent use of antibiotics in clinical settings without a clear mechanism for assessing performance over time. To address this gap, several international bodies have developed guidance around AMR measurement. For instance, in 2015, the Transatlantic Taskforce on Antimicrobial Resistance (TATFAR) developed a measurement framework, which consisted of a set of performance indicators that aim to track progress towards building more effective ASPs and to identify best practices (Box 5.3). In 2019, the WHO published a new toolkit that provided additional guidance for designing and implementing ASPs in low- and middle‑income countries (LMICs). Similar to the TATFAR measurement framework, the WHO proposed a set of clearly defined performance indicators that aim to track progress across multiple dimensions of care, including the structure and process of care, as well as patient outcomes (WHO, 2019[20]).
Box 5.3. TATFAR measurement framework for hospital-based antimicrobial stewardship programmes
Many ASPs lack performance targets to track progress in the prudent use of antibiotics in clinical settings. In recognition, TATFAR published a measurement framework in 2015, which consisted of a set of AMR-relevant indicators to help facilitate a common understanding of best practices in ASPs. The framework was developed through an iterative process, involving consultations with a multidisciplinary expert group convened by the US Centres for Disease control (CDC) and the European Centres for Disease Control (ECDC) (TATFAR, 2015[21]).
The TATFAR measurement framework consists of 33 AMR-relevant indicators (Table 5.2). Combined, these indicators capture important dimensions of ASPs in multiple domains, including their feasibility, clinical importance and relevance for minimising the AMR burden. Of these indicators, 17 are core indicators that can be used to characterise various dimensions of ASPs and the remaining 16 are optional indicators.
Table 5.2. TATFAR core indicators for hospital-based ASPs
|
Policy domain |
Indicator |
|---|---|
|
Infrastructure |
1. Does your facility have a formal antimicrobial stewardship programme accountable for ensuring appropriate antimicrobial use? 2. Does your facility have a formal organisational structure responsible for antimicrobial stewardship (e.g. a multidisciplinary committee focused on appropriate antimicrobial use, pharmacy committee, patient safety committee or other relevant structure)? 3. Is an antimicrobial stewardship team available at your facility (e.g. greater than one staff member supporting clinical decisions to ensure appropriate antimicrobial use)? 4. Is there a physician identified as a leader for antimicrobial stewardship activities at your facility? 5. Is there a pharmacist responsible for ensuring appropriate antimicrobial use at your facility? 6. Does your facility provide any salary support for dedicated time for antimicrobial stewardship activities (e.g. percentage of full-time equivalent for ensuring appropriate antimicrobial use)? 7. Does your facility have the information technology (IT) capability to support the needs of antimicrobial stewardship activities? |
|
Policy and practice |
8. Does your facility have facility-specific treatment recommendations based on local antimicrobial susceptibility to assist with antimicrobial selection for common clinical conditions? 9. Does your facility have a written policy that requires prescribers to document an indication in the medical record or during order entry for all antimicrobial prescriptions? 10. Is it routine practice for specified antimicrobial agents to be approved by a physician or pharmacist in your facility (e.g. pre‑authorisation)? 11. Is there a formal procedure for a physician, pharmacist or other staff members to review the appropriateness of an antimicrobial at or after 48 hours from the initial order (post-prescription review)? |
|
Monitoring and feedback |
12. Has your facility produced a cumulative antimicrobial susceptibility report in the past year? 13. Does your facility monitor if the indication is captured in the medical record for all antimicrobial prescriptions? 14. Does your facility audit or review surgical antimicrobial prophylaxis choice and duration? 15. Are results of antimicrobial audits or reviews communicated directly with prescribers? 16. Does your facility monitor antimicrobial use by grammes defined daily dose (DDD) or counts days of therapy (DOT) of antimicrobial(s) by patients per day? Has an annual report focused on antimicrobial stewardship (summary antimicrobial use and/or practices improvement initiatives) been produced for your facility in the past year? |
Source: TATFAR (2015[21]), Modified Delphi Process for Common Structure and Process Indicators for Hospital Antimicrobial Stewardship Programs, https://www.cdc.gov/drugresistance/pdf/summary_of_tatfar_recommendation_1.pdf.
The effectiveness of many ASPs can be enhanced by addressing the existing gaps in antibiotic guidance. For instance, in the United States, significant efforts have been made in recent years to provide antibiotic guidance for nursing homes, outpatient care and hospitals. Yet, one recent study found that about 28% of the outpatient antibiotic prescriptions filled for medication patients from 2004 to 2013 could not be linked with a record of a clinical encounter with a health worker in the previous week (Fischer et al., 2020[22]). Despite this, about half of the non-visit-based prescriptions had claims associated with laboratory tests or home care services. These results suggest that some prescribers may be responding to results obtained from tests or calls from home care services without having a clinical encounter with their patients.
Alternatively, extending guidance for relatively new modes of healthcare delivery can help improve the effectiveness of the existing ASPs. For instance, many ASPs lack guidance for antibiotic prescription during telehealth consultations, a relatively novel mode of healthcare delivery that gained popularity in the context of the ongoing COVID‑19 pandemic (Webster, 2020[23]). While the analytical base for the effectiveness of interventions that embedded telehealth services in the existing ASP guidelines is limited, emerging evidence offers promising results. For instance, the rollout of a telehealth-based ASP in 2 community hospitals in the United States was associated with a 24% decline in the prescription of broad-spectrum antibiotics within a 6‑month time frame (Shively et al., 2019[24]). In this period, consultations between local pharmacists and infectious disease physicians rose by 40.2% and the intervention led to savings on antimicrobial expenses. Another study from Brazil found that integrating telemedicine in an existing ASP led to a 30‑percentage point increase in the rate of appropriate antimicrobial prescribing (dos Santos et al., 2018[10]). This study also found significant declines in the use of fluoroquinolones, first-generation cephalosporins, vancomycin and polymyxins, as well as significant reductions in the rate of carbapenem-resistant Acinetobacter spp. Isolation (dos Santos et al., 2018[10]).
In countries where the prevalence of informal healthcare providers is high, an important policy priority is to address antibiotic prescription outside of formal clinical settings. In many OECD countries and key partners, antibiotics can only be prescribed by licensed health workers with formal medical education. Yet, in many LMICs, informal providers are an important source of healthcare. For instance, in India, an important global hotspot for AMR, informal providers without formal medical training represent a substantial fraction of the healthcare workforce. Much like many healthcare professionals with formal training, informal providers have been shown to rely frequently on antibiotics. For instance, one recent study from the West Bengal state found that, in nearly half of standardised patient interactions, informal providers prescribed antibiotics and about 70% of these prescriptions were unnecessary or harmful medicines (Das et al., 2016[25]).
Supporting prescribers’ decision making

Policy interventions
Computerised decision support systems and mobile health solutions.
Feedback interventions.
E‑prescribing.
Key messages
Computerised decision support tools (CDSTs) improve access to accurate antibiotic information relevant to prescribers’ decisions around dose optimisation and de‑escalation while facilitating AMR surveillance.
Mobile health technologies promote greater compliance with antibiotic guidelines.
Feedback interventions, including audits, real-time feedback and peer comparisons, encourage the prudent use of antibiotics.
E‑prescribing systems can enhance the quality of medical records that are used to inform the design and implementation of interventions to optimise prudent use of antibiotics.
Using computerised decision support systems and mobile health solutions
CDSTs can help optimise antibiotic use by accelerating access to accurate information at the time of prescription. In doing so, CDSTs can aid prescribers’ decisions around antibiotics including dose optimisation and de‑escalation. Systematic reviews focusing on CDSTs suggest that these technologies can spur greater compliance with antibiotic guidelines, with the effect size ranging from 6.9% to 61% (Curtis, Al Bahar and Marriott, 2017[26]). In community settings, significant improvements in antibiotic prescribing behaviours were documented in the treatment of acute otitis media (Holstiege, Mathes and Pieper, 2014[27]). Similarly, in hospital settings, one meta‑analysis by Curtis and colleagues (2017[26]) found that the use of CDSTs is associated with around twice as much appropriateness of antibiotic prescribing in line with the existing ASP guidelines (OR = 2.11, 95% CI, 1.67 to 2.66) (Curtis, Al Bahar and Marriott, 2017[26]).
Evidence is mixed in terms of the impact of CDSTs on antibiotic prescribing patterns as well as the use of healthcare resources. For instance, one study found that the rollout of the CDSTs in an intensive care unit was linked with reductions in antibiotic use, which was mirrored in the declines in the fraction of patients who were prescribed carbapenems, third-generation cephalosporins and vancomycin (Thursky et al., 2006[28]). This study also found a rise in the de‑escalation to narrower spectrum antibiotics. In comparison, one study from the United States found conflicting results in prescribing behaviours following the rollout of a CDST tool, with an 11.1% decline in intravenous defined daily dose and an accompanying 3.5% rise in oral defined daily dose (Fischer et al., 2003[29]). The evidence remains mixed in terms of the effects of CDSTs on the length of hospital stay and antimicrobial expenditures (Curtis, Al Bahar and Marriott, 2017[26]).
Mobile health technologies offer another avenue for improving access to accurate information at the point of care (Box 5.4). For instance, in New Zealand, one study used a mobile application that mapped the existing antibiotic guidelines directly to the prescribers’ mobile devices (Yoon et al., 2019[30]). This study found that the use of mobile applications was associated with an 8% increase in compliance with antibiotic guidelines for treating adult patients with community-acquired pneumonia. In Brazil, the rollout of a similar mobile application led to notable changes in prescribing patterns, with increases in the consumption of the recommended antibiotics like cefepime and concurrent declines in the use of piperacillin/tazobactam and meropenem (Tuon et al., 2017[31]).
Box 5.4. Supporting prescribers’ decision making through mobile technologies in the United Kingdom: Imperial Antibiotic Prescribing Policy (IAPP) application
In 2011, the IAPP application (app) was launched in a network of teaching hospitals within the overall organisation of the Imperial College Healthcare Trust, with the aim of ensuring prescribers’ access to antimicrobial prescribing policies at the point of care by replacing a physical pocket guide (Charani et al., 2017[32]). The IAPP app was developed through a collaborative process that involved the academic and clinical staff across Imperial College and took into account the prevalence of smartphone use among health workers (Charani et al., 2017[32]).
Importantly, a multimodal ASP was already in place in the participating hospitals prior to the launch of the app including (Charani et al., 2017[32]): i) dedicated multi-professional teams and pharmacists providing advisory services; ii) multidisciplinary ASP stewardship ward rounds; iii) a pocket guide that described all policy options distributed to all junior physicians upon start of employment and links to hospital intranet; iv) dedicated IPC control teams comprised of infection control nurses; v) a multidisciplinary Antibiotic Review Group responsible for developing and updating guidelines for antimicrobial use and infection treatment; and vi) activities around AMR awareness, education and feedback.
The IAPP was rolled out in a context where compliance with ASP guidelines was already high. Nonetheless, the introduction of the IAPP led to further increases in compliance in the tune of 6.48‑6.63% across different medical departments (Charani et al., 2017[32]). Importantly, the level of improved compliance was sustained over a 12‑month period. However, available evidence also suggests that the IAPP may have had unintended consequences, as the rollout of IAPP was associated with reductions in the fraction of prescriptions with a stop/review date, as well as declines in the documentation of indication (Charani et al., 2017[32]).
Source: Charani, E. et al. (2017[32]), “Effect of adding a mobile health intervention to a multimodal antimicrobial stewardship programme across three teaching hospitals: an interrupted time series study”, https://doi.org/10.1093/jac/dkx040.
Feedback interventions
A range of feedback interventions, including audits, real-time feedback and peer comparisons, can promote the prudent use of antibiotics. A key advantage of feedback interventions is that they can facilitate a rapid assessment of the existing challenges in prescription behaviours. Ideally, audits are carried out by ASP teams, which may be comprised of infectious disease physicians, clinical microbiologists and clinical pharmacists (Chung et al., 2013[33]). While audits can be carried out prospectively and retrospectively, the WHO recommends prospective audits whenever possible (WHO, 2019[20]). Real-time feedback can also be provided by ASP teams during ward rounds in oral or written form for either all inpatients or patients staying in high-risk areas like the intensive care unit. This feedback can subsequently be used for optimising antibiotic use (e.g. dose optimisation, intravenous (IV)-to‑oral switch).
Audits and real-time feedback interventions lead to improvements in compliance with antibiotic guidelines and reduce the use of hospital resources. One systematic review found that feedback interventions were commonly used in ASPs in paediatric care in hospital and outpatient settings and these interventions were associated with increases in compliance with antibiotic guidelines among physicians and attributable declines in the cost of treatment (Donà et al., 2020[34]). Another systematic review found that embedding feedback interventions into persuasive strategies used in ASPs may improve antibiotic prescribing behaviours in line with antibiotic guidelines (Davey et al., 2017[14]). Concerning hospital resources use, another systematic review found that these interventions were associated with 1 to 3.7‑day declines in the number of antibiotic treatment days in the intensive care unit (Van Dijck, Vlieghe and Cox, 2018[15]).
Peer comparison interventions can induce behaviour change among prescribers by increasing their awareness of their own antibiotic prescribing patterns in comparison to their peers (Navathe and Emanuel, 2016[35]). For instance, one cluster-randomised trial from the United States examined the impact of an ASP in primary care settings that compared an individual paediatrician’s prescribing performance against the performance of all paediatricians in the same primary care network (Gerber et al., 2013[36]). This study led to a 12.5 percentage point decline in broad-spectrum antibiotic prescribing in the intervention group. Another cluster-randomised trial among primary care practices in Boston and Los Angeles compared the antibiotic prescribing performance of clinicians to the top performers defined as prescribers with the lowest rates of inappropriate prescription rates (Meeker et al., 2016[37]). This intervention resulted in a 16.3 percentage point decline in the inappropriate prescription of antibiotics for acute respiratory tract infections. Another study from Norway demonstrated that feedback interventions can yield beneficial results even in settings with low AMR (Høgli et al., 2016[38]).
Importantly, feedback interventions can promote greater compliance with existing ASP guidelines by easing concerns over provider autonomy. One recent systematic review pointed out that several studies on ASPs raised concerns over the potentially adverse effects of restrictive strategies in service provision (Davey et al., 2017[14]). These potentially adverse effects include changes in professional culture due to difficulties that may arise from a breakdown in communication and trust between infection specialists and clinical teams (Davey et al., 2017[14]). In line with this review, available evidence suggests that if prescribers perceive ASPs as encroaching on their autonomy as providers, compliance with ASP guidelines may be lowered (Zetts et al., 2020[39]). This was the case in one 2015 study in the Lorraine region in France, which found that 68% of family physicians disfavoured restrictive interventions, which would require physicians to provide a justification for why the antibiotic that they prescribed complied with the existing ASP guidelines (Giry et al., 2016[40]). In recognition, some feedback interventions explicitly involve strategies to address concerns over provider autonomy. For instance, in the Netherlands, one hospital-based feedback intervention was designed specifically to preserve provider autonomy in an ASP by designating one provider in each department as the lead for good antibiotic policies, rather than requiring pre‑authorisation for antibiotics prescribed (Sikkens et al., 2017[41]). Physicians could, then, consult with their designated colleagues before they prescribed antibiotics (Sikkens et al., 2017[41]). This intervention resulted in significant increases in the appropriate use of antibiotics but the overall volume of antibiotic consumption remained the same.
Electronic prescribing (e‑prescribing)
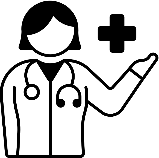
Ensuring high-quality medical record keeping is crucial for ensuring interventions that support the decision making of prescribers are built on accurate data. Despite this, keeping high-quality medical records remains a challenge in many countries (Figure 5.2). For instance, one recent EU point prevalence survey found that nearly 20% of medical records aggregated from 28 EU/EEA countries and Serbia did not provide any explanations for prescribing an antimicrobial (Plachouras et al., 2018[42]). In Australia, an analysis of the 2015 National Antimicrobial Prescribing Survey found that about 20% of antimicrobials lacked documented indication (NCAS/ACSQHC, 2016[43]). A subsequent study from Australia further showed that the share of prescriptions that lacked indication was lower among public hospitals that used e‑prescribing (8.4%) compared to hospitals that used paper-based systems (18.3%) (ACSQHC, 2021[44]). Another study from the United States documented that nearly 18% of all antibiotic prescriptions recorded in the 2015 National Medical Care Survey did not include a rationale for the prescription (Ray et al., 2019[45]).
Importantly, the quality of medical record keeping is linked with the type of antibiotics prescribed. One study from primary care settings in the United Kingdom found that the quality of documentation was the highest for frequently used first-line antibiotics and the poorest for infrequently used antibiotics (Dolk et al., 2018[46]). Another study from the United States showed that the likelihood of antibiotic prescriptions with incomplete information was the lowest for penicillin (Ray et al., 2019[45]). This study also showed that the likelihood of incomplete antibiotic prescription was the lowest for penicillin. A similar variation in antibiotic documentation quality was highlighted in a recent review of 27 lower-middle‑income countries (LMICs) (Sulis et al., 2020[47]).
E‑prescribing enhances the quality of medical record keeping and supports efforts to monitor antibiotic use in health facilities. Earlier systematic reviews found that e‑prescribing is associated with reductions in medication errors and the risk of adverse drug events (Ammenwerth et al., 2008[48]). In line with these findings, a subsequent recent meta‑analysis concluded that e‑prescribing interventions were associated with a 76% reduction in medical errors (Relative risk = 0.24 [95% CI 0.13, 0.46]), dosing errors (Relative risk = 0.17 [95% CI 0.08, 0.38]) and adverse drug events (Relative risk = 0.52 [95% CI 0.40, 0.68]), though no statistically significant effects were observed for length of hospital stay or for mortality (Roumeliotis et al., 2019[49]). However, these findings should be interpreted with caution because the quality of evidence used in these analyses was determined to be very low (Roumeliotis et al., 2019[49]).
Figure 5.2. Share of medical records with no clear documentation of indication for antimicrobial prescription, various years
Source: China: nationally representative database (Beijing Data Centre for Rational Use of Drugs) covering the years 2014‑18 as published in Zhao, H. et al. (2021[50]), “Appropriateness of antibiotic prescriptions in ambulatory care in China: A nationwide descriptive database study”, https://doi.org/10.1016/s1473-3099(20)30596-x; United States: taken from 2015 National Ambulatory Medical Care Survey as published in Ray, M. et al. (2019[45]), “Antibiotic prescribing without documented indication in ambulatory care clinics: National cross sectional study”, https://doi.org/10.1136/bmj.l6461; 28 EU/EEA countries and Serbia: data aggregated and extracted from the 2016‑17 second point prevalence survey (PPP) of healthcare‑associated infections as reported in Plachouras, D. et al. (2018[42]), “Antimicrobial use in European acute care hospitals: Results from the second point prevalence survey (PPS) of healthcare‑associated infections and antimicrobial use, 2016 to 2017”, https://doi.org/10.2807/1560-7917.es.23.46.1800393; Australia: extracted from the 2015 National Antimicrobial Prescribing Survey as published in NCAS/ACSQHC (2016[43]), Antimicrobial Prescribing Practices in Australian Hospitals: Results of the 2015 Hospital National Antibiotic Prescribing Survey, National Centre for Antimicrobial Stewardship and Australian Commission on Safety and Quality in Health Care.
Moreover, e‑prescribing systems are shown to contribute to ongoing ASP interventions by providing facility-level data that can be used for monitoring and improving antibiotic use. A potential benefit of e‑prescribing is that information on antimicrobial use is recorded regularly in these systems, which can serve as novel data sources to assess and monitor antimicrobial use in health facilities and inform the design of interventions to improve antibiotic prescribing behaviours.
One recent systematic review found that the use of e‑prescribing systems for quantitative data analysis remains limited, though in some OECD countries, efforts are being made to incorporate data generated from e‑prescribing systems into the existing ASPs (Micallef et al., 2017[51]). For instance, several studies from Australia used e‑prescribing data to support auditing and feedback interventions focusing on antimicrobial prescribing behaviours of doctors (Micallef et al., 2017[51]). In the United States, data from e‑prescribing systems that track antimicrobial dispensing volumes, course durations and doses have been utilised to evaluate the impact of AMR policies. Other studies from Germany, South Korea and the United States use these data for quality improvements (Micallef et al., 2017[51]).
Pharmaceutical policies
Policy interventions
Promoting the use of forgotten antibiotics.
Separating antibiotic prescribing from dispensing.
Key messages
Removing economic and regulatory barriers to the market registration of forgotten antibiotics can help enhance access to these antibiotics.
Addressing the shortages in medicines can ensure adequate access to forgotten antibiotics.
Promoting local and global collaborations can help accelerate access to forgotten antibiotics.
Separate prescription and dispensing of antibiotics can lower the overall volume of antibiotic prescription.
Promoting the use of forgotten antibiotics
One pharmaceutical intervention that can help curb the unnecessary use of antibiotics is promoting the use of forgotten antibiotics. Forgotten antibiotics refer to a class of older antibiotics that can be used in treating patients even though they became available decades earlier. Despite their potential benefits, many OECD countries, EU/EEA and G20 countries do not frequently rely on delayed antibiotic prescribing, with only around a quarter of action plans to tackle AMR making any references to these antibiotics.
Reducing economic and regulatory barriers to market registration is key to improving access to forgotten antibiotics. Many older antibiotics reflect the quality standards in clinical trials and requirements for regulatory documentation at the time of entry into the pharmaceutical market. This can act as a barrier against the increased use of forgotten antibiotics because registering these antibiotics will entail renewed data collection efforts and additional expenses for drug registration, whereas pharmaceutical companies may not always have incentives for registering these antibiotics in new markets (Cox et al., 2015[52]; WHO, 2018[53]). Easing the economic and regulatory barriers to their market registration is one option to increase access to forgotten antibiotics (Pulcini et al., 2017[54]).
In recent years, several laudable examples of co‑operation and collaboration across national and international agencies, academic institutions and pharmaceutical have emerged to accelerate market authorisation of new drugs for treating multidrug-resistant tuberculosis (Box 5.5). Lessons learnt from these examples can be applied to ease the regulatory barriers that hinder the entry of forgotten antibiotics into the pharmaceutical market (Cox et al., 2015[52]; Pulcini et al., 2017[54]; WHO, 2018[53]).
Box 5.5. Accelerating access to new drugs for treating multidrug-resistant tuberculosis
One recent example of international efforts to accelerate the introduction of new drugs into the pharmaceutical markets involves antibiotics used for treating multidrug-resistant tuberculosis: bedaquiline and delamanid. These two drugs are currently used for treating tuberculosis patients with bacteria resistant to multiple drugs including rifampicin and isoniazid – two crucial drugs used in the treatment of tuberculosis (WHO, n.d.[55]). Prior to the advent of these drugs, the last time a new drug was developed specifically to treat tuberculosis was when rifampicin was rolled out in the 1960s (WHO, n.d.[55]). In recent years, resistance to rifampicin has been reported across the globe (WHO, 2017[56]).
In recognition of growing concerns over the emergence of multidrug-resistant tuberculosis, the market approval of these two drugs was accelerated. In 2012, the US Food and Drug Administration provided conditional market approval for the use of bedaquiline (WHO, 2017[56]). Following this approval, the WHO published interim guidance in 2013 for its use for patients suffering from multidrug-resistant tuberculosis (WHO, 2013[57]). The following year, in 2014, delamanid received conditional market authorisation from the European Medicine Agency Committee for Medicinal Products for Human Use. Subsequently, the WHO published interim guidance to assist countries in their efforts to ensure that the treatment regimens are taken up in a safe and effective manner (WHO, 2017[56]).
Source: WHO (n.d.[55]), Frequently Asked Questions on Bedaquiline; WHO (2013[57]), The Use of Bedaquiline in the Treatment of Multidrug-resistant Tuberculosis. https://apps.who.int/iris/handle/10665/84879; WHO (2017[56]), “WHO best-practice statement on the off-label use of bedaquiline and delamanid for the treatment of multidrug-resistant tuberculosis, https://apps.who.int/iris/handle/10665/258941.
In addition to easing market access for these antibiotics, addressing the shortages in existing medicines is another strategy that can help alleviate the challenges in access to forgotten antibiotics. Many forgotten antibiotics are categorised as Access antibiotics in accordance with WHO AwARE groupings (see Chapter 3) that can be used for treating common infections (WHO, 2021[58]). The WHO guidance indicates that Access antibiotics should be accessible at affordable prices and in a quality-assured manner. Despite this, many OECD countries are reporting antibiotic shortages. For example, nearly all pharmacists (95%) that participated in a 2019 pan‑European survey from 39 countries indicated that the shortage of medicines was a major problem in the hospital where they worked, a marked rise from about 86% in 2014 (EAHP, 2019[59]). An earlier wave of this survey reported that the average duration of a typical medicine shortage was around 2.2 months in 2018, suggesting that these shortages can result in delays and cancellations in needed care or therapy and receiving a suboptimal course of care. A closer look at the 2019 survey shows that antimicrobial agents are the leading cause of shortages in medicines since 2014. In 2019, about 63% of pharmacists indicated that they experienced shortages in antimicrobial agents, a significant increase compared to 57% in 2014 (EAHP, 2019[59]). While the precise impact of these shortages on patient outcomes is largely unknown, it is recognised that these shortages are a global health priority and a barrier to tackling AMR.
Separating prescription and dispensing of drugs
In most OECD countries, antibiotics are prescribed by health workers and dispensed separately from pharmacies, with no financial links between the prescriber and the dispensing pharmacy. But certain exceptions exist. For instance, in the United Kingdom, nearly 13% of practices in the National Health Service primary care have an inhouse dispensary (Goldacre et al., 2019[60]). These types of dispensing practices are primarily located in rural areas with a lower density of pharmacies, thus providing necessary access to patients residing in these areas (Goldacre et al., 2019[60]). In Switzerland, drug dispensing is regulated by each state, referred to as cantons. While some cantons apply a strict separation between prescribing and dispensing of drugs, others either have no separation or a mixed system (Trottmann et al., 2016[61]).
Evidence from countries where health workers are allowed to dispense the antibiotics that they prescribed to their own patients suggests that there are linkages between physicians’ dispensing responsibilities and prescribing behaviours. For instance, in the United Kingdom, dispensing primary care practices were more likely to prefer drugs with higher costs across all classes (Goldacre et al., 2019[60]). In Switzerland, one study found that the likelihood of per capita antibiotic consumption was around 1.3 times higher in areas where more than half of the practitioners dispensed drugs directly to their patients (Filippini, Heimsch and Masiero, 2014[62]). Another study from Switzerland found that physician dispensing was associated with a switch towards generic drugs over brands (Trottmann et al., 2016[61]). This study further showed that the greater reliance on generic drugs led to lower pharmaceutical spending for each patient, which was offset by increased use of physician services. Conversely, in Australia, one earlier study found that physician dispensing was associated with fewer drug prescriptions, which was partly explained by perceived expectations from their peers about their prescribing behaviours and preference to generate less administrative paperwork (Lim et al., 2011[63]).
Experiences from countries that implemented separation policies suggest that these interventions can be effective in spurring declines in overall antibiotic prescription and improvements in prudent use. One relatively well-documented example of a separation policy comes from South Korea. In 2002, South Korea put in place a new separation policy, which outlawed prescribing doctors from dispensing drugs and pharmacists from prescribing (Park et al., 2005[64]). A recent evaluation found that this policy was associated with reductions in antibiotic prescriptions for patients with viral illnesses (Relative risk = 0.89, [95%CI: 0.86, 0.91) and to a smaller extent for patients with bacterial illness (Relative risk = 0.98, [0.97, 0.99]) (Park et al., 2005[64]). This study further concluded that the separation policy resulted in the decline of inappropriate antibiotic prescriptions for viral illnesses.
Country experiences suggest that separating the prescription and dispensing of drugs may have unintended consequences. For instance, a recent study suggested Korea’s separation policy may have contributed to increases in medical expenditures, partly because the reform led to substantial increases in service fees for providers to offset the potential loss of income and resulted in shifts in prescribing patterns that favoured brand-name or imported drugs (Kim and Ruger, 2008[65]).
Information-based strategies

Policy interventions
AMR awareness campaigns.
Improving health literacy.
Key messages
AMR awareness campaigns should ensure to have clear public health messaging to dispel confusion and misconceptions about antibiotic use.
Improving the health literacy of the general population promotes more prudent use of antibiotics.
AMR awareness campaigns
As discussed in Chapter 4, many OECD, EU/EEA and G20 countries often rely on mass media campaigns to raise awareness around AMR in the general public but available evidence suggests that these efforts have yielded modest effects. This finding should be interpreted with care, however, as the majority of evidence is generated through studies with weak methodologies. To date, several studies examined the effectiveness of AMR awareness campaigns targeting the general public. In Italy, one AMR awareness campaign aimed at improving the antibiotic behaviours of patients, specifically for antibiotics to treat upper respiratory infections. One study that evaluated the impact of this campaign through a non‑randomised trial found that antibiotic prescribing was reduced by 4.3% in the intervention area in comparison to the control area (Formoso et al., 2013[66]). In the United Kingdom, one study recently examined the impact of a regional mass media campaign that was implemented for two consecutive years using editorial coverage from local newspapers, television and radio stations. This study found that there was a 5.8% reduction in antibiotic prescription in the intervention area in comparison to the control area (Lambert, Masters and Brent, 2007[67]). In the United States, one community-wide intervention in the state of Tennessee targeted healthcare providers that routinely provided healthcare services for children, parents of young children and the general public. Parent education activities consisted of the distribution of pamphlets and public education activities included the distribution of pamphlets and dissemination of information on television, radio stations, newspapers and public service announcements. The majority of the educational materials were developed by the CDC (Perz, 2002[68]). This intervention was associated with an 11% decline in antibiotic prescription for young children compared to the control communities that did not roll out similar awareness campaigns in the study period.
One potential explanation for the observed modest effects is that confusion and misconception about antibiotics may impede behaviour change in settings where antibiotic knowledge is already relatively high. A recent WHO survey of 12 LMICs across all 6 WHO regions showed that most respondents (64%) understood that AMR was a significant challenge with consequences for themselves and their families (WHO, 2015[69]). But most respondents lacked an understanding of the potential channels through which AMR affects their own lives and what they could personally do to address it. Confusion around illness types that can be treated with antibiotics was also common. Similar misconceptions and confusion around resistant pathogens and transmission channels have also been documented in high-income settings, such as Australia (Bakhit et al., 2019[70]), Italy (Prigitano et al., 2018[71]) and the United States (CIDRAP, 2019[72]).
To tackle confusion and misconceptions, AMR awareness campaigns should rely on clear public health messaging. A recent review of large‑scale antibiotic awareness campaigns from 93 countries since 2010 concluded that the effectiveness of these efforts is often hindered by unclear public health messages around AMR (Huttner et al., 2019[73]). Moreover, this study suggested that AMR awareness campaigns should update their key messages regularly and involve experts with backgrounds in healthcare, health communication and social marketing to strengthen their implementation. This was the case in Italy, where the AMR awareness campaign discussed earlier tailored the key messages of the campaign based on consultations with physicians from various health districts in an attempt to understand the unique contextual factors in the area of their practice that may shape the attitudes and expectations of patients around antibiotics (Formoso et al., 2013[66]).
Improving health literacy
Individuals with greater levels of health literacy may have better access to health information, as well as a greater ability to process and act on this information in a way that promotes prudent antibiotic use. For instance, in Germany, one study found that people with sufficient health literacy, measured through a 16‑point scale, were almost half as likely to have a recent history of antibiotic use than those with insufficient health literacy (Salm et al., 2018[74]).
Despite increasing recognition of its importance, health literacy remains low in many OECD countries. One recent survey found that only about half (52.5%) of participants from eight European countries had sufficient or excellent health literacy scores (Sørensen et al., 2015[75]). This study also concluded that people with low socio‑economic status and educational attainment and older adults faced greater deficits in health literacy. More recently, one OECD working paper also found that a considerable proportion of respondents experienced difficulty in evaluating the reliability of the information provided in the media (47%), weighting the advantages and disadvantages of different treatment options (41%) and deciding whether they need vaccines (31%) (Moreira, 2018[76]). The OECD members use a wide range of policies to improve the health literacy of their populations, including: the promotion of health literacy skills among adults and children; counselling and training activities in community settings; guidelines aimed at enhancing health professionals’ communication skills; and easing access to health information (Moreira, 2018[76]).
Policies to prevent or reduce the emergence of resistant infections
IPC programmes at the national and health facility levels
The WHO IPC guidelines indicate that IPC programmes are most effective when they combine strategies that: i) promote the right mix of health professionals with adequate IPC training; ii) improve staff workload, bed capacity and physical attributes of health facilities; iii) enhance the accessibility of equipment and supplies; and iv) promote a work culture that enables effective IPC practices (WHO, 2016[77]). Importantly, these efforts are meant to be complemented by IPC surveillance, monitoring and feedback practices at the local and national levels. In recent years, many technical tools have been developed in support of the 2016 WHO IPC guidelines (Storr et al., 2017[78]; WHO, 2018[79]).
Policy interventions
Integrating AMR in healthcare‑associated infection (HAI) surveillance.
IPC monitoring, evaluation and feedback.
Dedicated IPC leadership in health facilities.
Optimising organisation of healthcare delivery.
Key messages
Integrating AMR in HAI surveillance facilitates systematic data collection and analysis.
Building dedicated IPC teams helps monitor ongoing IPC practices, educate health workers and promote a work environment that enables the best IPC practices.
Scaling up IPC monitoring, regular audits, evaluation and feedback interventions can promote greater compliance with IPC guidelines among health workers.
Addressing high rates of bed occupancy and overcrowding in health facilities can help reduce the likelihood of healthcare‑acquired resistant infections.
Integrating AMR in HAI surveillance
HAI surveillance can facilitate standardisation in the collection and analysis of data over time. For instance, one recent review of 42 HAI surveillance systems across 20 European countries and 4 transnational systems showed that about 64.2% of these surveillance systems track the percentage of resistant isolates to specific drugs (Núñez-Núñez et al., 2018[80]).
AMR surveillance facilitates the identification of patterns of AMR pathogens in healthcare settings. One study from an Italian teaching hospital used data from a prospective HAI surveillance programme for a period of years (Bianco et al., 2018[81]). This study concluded that the most common resistant pathogens were Gram-negative bacteria, including Klebsiella pneumoniae (K. pneumoniae), Acinetobacter baumannii, Escherichia coli (E. coli) and Pseudomonas aeruginosa (P. aeruginosa). In Canada, one study used a 4‑year time series of surveillance data collected from 70 sentinel hospitals that participated in the Canadian Nosocomial Infection Surveillance Program (CNISP/Public Health Agency of Canada, 2020[82]). This study found that infection rates for MRSA and vancomycin-resistant enterococci bloodstream infections increased by 59% and 143% respectively. In contrast, Clostridioides difficile (C. difficile) infection rates declined by 12.5% from 2015 to 2018.
The effectiveness of HAI surveillance can be enhanced by adopting automated surveillance systems that track a comprehensive range of infections. Manual review of patient charts can be labour-intensive and lack standardisation (Streefkerk et al., 2020[83]). In recognition, many countries are increasingly using automated surveillance systems either to support the existing manual surveillance strategies (i.e. semi‑automated surveillance) or replace them altogether (van Mourik et al., 2017[84]). Automated surveillance systems can target a specific set of infections (e.g. infections observed in intensive care units, surgical site infections and device‑associated infections) or they can be used for comprehensive surveillance (Streefkerk et al., 2020[83]). However, evidence suggests that targeted surveillance can also miss important HAIs, as is demonstrated by one study from the United States which quantified that targeted surveillance can miss up to half of all HAIs (Weber et al., 2012[85]).
IPC monitoring, evaluation and feedback
Regular IPC audits and feedback significantly improves compliance with IPC guidelines. For instance, one cluster-randomised experiment showed that using daily audits with regular feedback and a checklist that clearly identified the priority process indicators was associated with increased compliance with recommended IPC guidelines (Charrier et al., 2008[86]). Alternatively, peer assessments with anonymous feedback were also shown effective in improving compliance with handwashing guidelines (Storr et al., 2017[78]). Another study from England and Wales (United Kingdom) demonstrated that personalised feedback with explicit goal-setting exercises was associated with a 10‑13% increase in compliance with hand hygiene guidelines in acute care for the elderly and 13‑18% in intensive care units (Fuller et al., 2012[87]).
Automated auditing techniques offer a promising avenue for improving compliance with IPC guidelines. In many settings, audit- and feedback practices rely on direct observation of behaviours but emerging evidence suggests direct observation can spur inaccuracies in data collection and create tension among health workers (Livorsi et al., 2018[88]). Automated auditing techniques can offer an alternative solution. For instance, one recent study showed that automatic video auditing with feedback led to a 15.7% to 46% increase in compliance with the WHO’s handwashing guidelines (Lacey et al., 2020[89]). Another study that used remote video surveillance with real-time group feedback led to similar increases in compliance with hand hygiene guidelines in an intensive care unit (Armellino et al., 2011[90]).
Dedicated IPC leadership at health facilities
Creating dedicated IPC teams and leaders in health facilities is crucial to promote best practices. Dedicated IPC teams can: monitor the ongoing IPC practices; educate and train other health professionals; and foster a work environment that promotes best practices. While the exact composition of the IPC teams will differ depending on the context of care, multidisciplinary teams comprised of nursing staff, a dedicated physician with training in infection control and other health personnel that can provide microbiological and data management support are preferred.
Dedicated IPC leadership should encourage and support the provision of IPC training and education, particularly in the context of health emergencies. The beneficial impact of IPC training and education is well-documented (Zingg et al., 2015[91]; Storr et al., 2017[78]). In recent years, a growing body of evidence shows that simulation-based IPC training is associated with reductions in HAIs (Wang et al., 2019[92]), including central line‑associated bloodstream infections (Allen et al., 2014[93]; Gerolemou et al., 2014[94]). Moreover, in the context of health emergencies, these benefits may be pronounced. One recent systematic review showed that the provision of IPC training and education was consistently linked to reduced risk of infections among health workers, not only in the context of the ongoing pandemic but also during SARS‑CoV‑1 and MERS-CoV outbreaks (Chou et al., 2020[12]).
IPC teams and leaders in acute care facilities should also support clear communication of IPC guidelines and promote a work environment that promotes the best IPC practices. The recent Cochrane review provided evidence that beliefs and attitudes among health professionals towards IPC practices shape the professional culture and practices in health facilities (Houghton et al., 2020[95]). This review further showed that health professionals had difficulty complying with local IPC guidelines when these guidelines were lengthy and offered confusing explanations of the recommended code of IPC practice (Houghton et al., 2020[95]). Moreover, inconsistencies across local, national and international IPC guidelines and frequent updates further undermined the likelihood of compliance. Combined, evidence suggests that dedicated IPC teams can support efforts to clearly communicate IPC guidelines through education and training activities. To date, available evidence showed that IPC training and education programmes yielded improvements in provider knowledge of IPC practices, led to gains in provider competency and enhanced compliance with existing IPC guidelines as much as 27.5% (Storr et al., 2017[78]; Wang et al., 2019[92]).
Optimising the organisation of care
Keeping the workload of health workers at acceptable levels is key to avoiding increases in the likelihood of healthcare‑acquired AMR even in settings where IPC guidelines are in place and compliance is high. One cohort study from Portugal concluded that the quality of antimicrobial prescriptions was inversely related to the workload of the prescribing physician (Teixeira Rodrigues et al., 2016[96]). Similarly, a more recent study from China showed that reductions in the workload of prescribing physicians were inversely associated with the rate of carbapenem-resistant Pseudomonas aeruginosa (Han and Zhang, 2020[97]).
Addressing high rates of bed occupancy and overcrowding is another important intervention to reduce the risk of resistant HAIs. Several recent studies demonstrated that high rates of bed occupancy and overcrowding contribute to the spread of MRSA by: decreasing compliance with IPC guidelines (e.g. hand hygiene); increasing movement of patients and healthcare staff between different wards within health facilities; decreasing levels of cohorts; and overburdening available resources for screening and isolation (Andersen et al., 2002[98]; Clements et al., 2008[99]). Similarly, a recent study from the United States showed that a 1% increase in private patient rooms as a proportion of all inpatient rooms was associated with an 0.8% decline in MRSA infections effect (Park et al., 2020[100]).
Among OECD members, the curative care bed occupancy rate remained relatively stable over the last decade, averaging at about 76% in 2019 (Figure 5.3). However, substantial variation exists across countries in the occupancy rates: in Canada, almost 92% of acute care beds were used in 2019 compared to 63.4% in the Netherlands. In addition to bed occupancy and overcrowding, certain attributes of the physical infrastructure (e.g. the lack of isolation rooms, shower facilities and difficulties in access to handwashing facilities, poor water, sanitation and waste management) and inadequate access to equipment and materials have been reported to hinder good IPC practices (Storr et al., 2017[78]).
Policies to improve vaccination coverage

Policy interventions
Addressing hesitancy towards vaccines.
Key messages
A wide range of communication and dialogue‑based interventions can be used to build and sustain public confidence vaccines among different stakeholders.
Behavioural interventions are showing promising results in nudging people to take up vaccines.
While it is paramount to support R&D efforts for developing new antibiotics and promote the use of forgotten ones, reliance on antibiotics will not suffice to mitigate the AMR threat. In contrast to antibiotics, vaccines are currently undergoing a remarkable phase of development and technological advances, offering new avenues for tackling AMR (Box 5.6). In recognition of the importance of vaccines for tackling AMR, the WHO Global Action Plan on Antimicrobial Resistance (GAP AMR) explicitly urges the global community to make an economic case for improved investments in vaccines.
Improving vaccination coverage is considered a public health priority in many countries. Building on long-standing efforts to improve vaccination coverage, in 2012, all members of the WHO endorsed the Global Vaccine Action Plan 2011‑20. This plan sets out a 90% coverage target for all vaccines globally by 2020, with each country achieving at least 80% of coverage within all of their subnational administrative units (WHO, 2012[101]). Since then, many countries have seen marked increases in the coverage of different vaccines, though this progress differs by country groupings. As of 2019, the Haemophilus influenzae type B (Hib) vaccine has been incorporated into routine immunisation programmes in 192 countries (IVAC, 2019[102]). In contrast, only less than half of all infants (47%) residing in 144 countries have access to pneumococcal conjugate vaccines (PCVs). In 2018, the PCV coverage was the highest in the WHO’s Americas and Europe regions: respectively, 82% and 78% of infants at 1 year of age received 3 doses of PCV in 2018 (WHO, 2019[103]). In the same year, the PCV coverage rates averaged 13% in the WHO Western Pacific region and 17% in the South-East Asia region (WHO, 2019[103]).
Box 5.6. New developments in new vaccine candidates to tackle AMR
In recent years, important strides have been made in the development of new vaccine candidates that can help reduce AMR. Developing a new vaccine can be a prolonged process, with only a few vaccine candidates eventually completing the clinical trial phase. Nevertheless, recent developments in vaccine research yielded promising results. For instance, a glycoconjugate vaccine that can protect against infections from P. aeruginosa and K. pneumoniae is currently in pre‑clinical development, with early evidence demonstrating promising results (Hegerle et al., 2018[104]). Both of these bacteria are catalogued as a priority by the WHO and have been increasingly shown to complicate existing strategies for treatment.
Another pathogen that has recently been shown to develop a multidrug-resistant strain, particularly in community settings, is hypervirulent K. pneumoniae. Recent pre‑clinical trials with a novel bioconjugate vaccine targeting hypervirulent K. pneumoniae have shown promising results in pre‑clinical studies (Feldman et al., 2019[105]). In addition to these efforts, a number of vaccine candidates are currently in different stages of clinical development, including those that target C. difficile, group B Streptococcus, Mycobacterium tuberculosis, Staphylococcus aureus (S. aureus), carbapenem-resistant and extra intestinal E. coli and respiratory syncytial virus (Buchy et al., 2020[106]).
A growing body of literature highlights the need for incorporating the potential benefits of vaccines for AMR to reflect their full value (Bloom et al., 2017[107]). For instance, one recent review of the mathematical models that inform the implementation of vaccination programmes concluded that only a handful of studies in the existing literature quantified the impact of a vaccine on antibiotic transmission within a human population (Atkins et al., 2018[108]).
Source: Hegerle, N. et al. (2018[104]), “Development of a broad spectrum glycoconjugate vaccine to prevent wound and disseminated infections with Klebsiella pneumoniae and Pseudomonas aeruginosa”, https://doi.org/10.1371/journal.pone.0203143; Feldman, M. et al. (2019[105]), “A promising bioconjugate vaccine against hypervirulentKlebsiella pneumoniae”, https://doi.org/10.1073/pnas.1907833116; Buchy, P. et al. (2020[106]), “Impact of vaccines on antimicrobial resistance”, https://doi.org/10.1016/j.ijid.2019.10.005; Bloom, D. et al. (2017[107]), “Moving beyond traditional valuation of vaccination: Needs and opportunities”, https://doi.org/10.1016/j.vaccine.2016.12.001; Atkins, K. et al. (2018[108]), “Use of mathematical modelling to assess the impact of vaccines on antibiotic resistance”, https://doi.org/10.1016/s1473-3099(17)30478-4.
In general, vaccination coverage among OECD members remains relatively high, but some countries are experiencing challenges in maintaining performance. On average, nearly 95% of children residing in OECD countries receive the recommended diphtheria, tetanus and pertussis (DTP) and measles vaccines, and around 91% receive a vaccine for hepatitis B (OECD, 2019[109]). Despite this, some OECD countries have seen reductions in the coverage of DTP vaccines by as much as 4 or more percentage points (e.g. Canada, Chile, Iceland, Lithuania, Mexico, Poland, Slovenia and Spain), while others experienced smaller declines (Estonia, Iceland, Lithuania, the Netherlands, Poland, the Slovak Republic and Slovenia) (OECD, 2019[109]). Today, nearly half of OECD countries do not meet the minimum immunisation thresholds (95%) recommended by the WHO to prevent the spread of measles and 15% fall short of meeting a similar target for DPT (OECD, 2019[109]). Taken together, these findings suggest that even countries with a long-standing track record of high vaccination coverage can face challenges in maintaining performance.
Addressing hesitancy towards vaccines
The remainder of this section focuses on barriers that may hinder the performance of vaccination programmes, with an emphasis on hesitations around vaccines. This approach was taken as a recognition of the growing concerns around hesitation towards the vaccines available among OECD members, despite generally high levels of vaccination coverage (Box 5.7).
Box 5.7. Vaccine hesitancy in OECD countries
Vaccine hesitancy can significantly threaten the progress made in tackling vaccine‑preventable diseases. Broadly, the WHO defines vaccine hesitancy as the reluctance and/or refusal to the vaccine, even though the vaccines are available. The WHO calls out vaccine hesitancy among the top ten gravest threats to global health. Vaccine hesitancy is a complex public health challenge due to its context-specific drivers that vary across time and different vaccine types (Díaz Crescitelli et al., 2020[110]; Larson et al., 2014[111]). Available evidence suggests that refusal of vaccines is on the rise among some OECD members, with the refusal rates for the measles-mumps-rubella (MMR) vaccine in 9 EU countries increasing over the last two decades (Larson et al., 2018[112]).
Waning public confidence in vaccines is among the most important factors that exacerbate hesitancy towards vaccines. In recent years, EU countries are diverging in terms of general public confidence in vaccines with some countries experiencing improvements (e.g. France, Italy and Slovenia), while others are seeing declines (e.g. the Czech Republic, Finland and Sweden) (Larson et al., 2018[112]). Importantly, vaccine confidence among health professionals also varies. For instance, in the Czech Republic, 36% of general practitioners indicated that they disagreed with the statement that the MMR vaccine was safe (Larson et al., 2018[112]).
Public confidence in vaccines is highly correlated with trust in the importance, safety and effectiveness of vaccines. One recent study that collated data across 149 countries found that, in 2020, among OECD countries and key partners, EU/EEA and G20 countries, the proportion of study participants that strongly believed that vaccines were important to child health stood around 67% (Figure 5.4) (de Figueiredo et al., 2020[113]). Importantly, considerable variation across countries exists in the beliefs about the importance of vaccines, ranging above 80% in Argentina, Brazil, China, Colombia, Costa Rica, Finland, Iceland, India and Romania, below 50% in Bulgaria, the Czech Republic, Israel, Japan, Korea, Sweden and Türkiye.
Figure 5.4. Beliefs about the importance of vaccines among OECD countries, 2020
Source: The graph presents the mean model-based estimates of the proportion of respondents who strongly agreed that vaccines are important for children for the year 2020 as published in de Figueiredo, A. et al. (2020[113]), “Mapping global trends in vaccine confidence and investigating barriers to vaccine uptake: A large‑scale retrospective temporal modelling study”, https://doi.org/10.1016/s0140-6736(20)31558-0, which generated estimates based on data from 290 surveys carried out from September 2015 to December 2019 across 149 countries.
Beliefs about the safety of vaccines also vary among G7 countries, OECD members and key partners (Figure 5.5). De Figueiredo et al. (2020[113]) also showed that, in 2020, only about 48% of study participants from OECD countries and key partners, EU/EEA and G20 countries considered vaccines to be safe. Beliefs about the safety of vaccines vary substantially, with a fraction of study participants indicating that they strongly agree with the statement that vaccines are important standing at the lowest levels for those Japan (18%) and Lithuania (19%), and the highest for those study participants from India (82%), Costa Rica (76%) and Argentina (76%). Similar variation in the beliefs about the effectiveness of vaccines was observed among G7 countries, OECD countries and key partners.
Figure 5.5. Beliefs about the safety of vaccines among OECD countries, 2020
Source: The graph presents the mean model-based estimates of the proportion of respondents who strongly agreed that vaccines are safe for children for the year 2020 as published in de Figueiredo, A. et al. (2020[113]), “Mapping global trends in vaccine confidence and investigating barriers to vaccine uptake: A large‑scale retrospective temporal modelling study”, https://doi.org/10.1016/s0140-6736(20)31558-0, which generated estimates based on data from 290 surveys carried out from September 2015 to December 2019 across 149 countries.
With respect to the beliefs about the effectiveness of vaccines (Figure 5.6), there is also substantial cross-country variation. It is estimated that, for the year 2020, about half of respondents (54%) in OECD countries and key partners, EU/EEA and G20 countries strongly agreed with the statement that vaccines are effective for children, with Japan having the lowest level of agreement (22%) and India having the highest level of agreement (85%) with this statement.
Figure 5.6. Beliefs about the effectiveness of vaccines among OECD countries, 2020
Source (box): Díaz Crescitelli, M. et al. (2020[110]), “A meta-synthesis study of the key elements involved in childhood vaccine hesitancy”, https://doi.org/10.1016/j.puhe.2019.10.027; Larson, H. et al. (2014[111]), “Understanding vaccine hesitancy around vaccines and vaccination from a global perspective: A systematic review of published literature, 2007‑12”, https://doi.org/10.1016/j.vaccine.2014.01.081; Larson, H. et al. (2018[112]), State of Vaccine Confidence in the EU, https://doi.org/10.2875/241099; de Figueiredo, A. et al. (2020[113]), “Mapping global trends in vaccine confidence and investigating barriers to vaccine uptake: A large‑scale retrospective temporal modelling study”, https://doi.org/10.1016/s0140-6736(20)31558-0.
Increasingly, several international bodies are publishing analytical products to guide country efforts to address vaccine hesitancy, which underscores the need to build and sustain public confidence in vaccines among different stakeholders. Recently, the WHO also made available best practice guidelines that aim to provide basic principles for promoting vaccines in public discussions (WHO/Europe, 2017[114]). In 2017, the European Centre for Disease Prevention and Control (ECDC) published a catalogue of individual- and community-level strategies to alleviate vaccine hesitancy (2017[115]). In accordance with emerging evidence, the majority of the strategies highlighted by the ECDC rely on communication and dialogue techniques to address the information-related drivers of vaccine hesitancy, including those that relate to lack of information, misinformation and mistrust (Díaz Crescitelli et al., 2020[110]). Further, they highlight the need for developing strategies that target the complex driver of vaccine hesitancy across a wide array of stakeholders, including parents, health professionals and community gatekeepers as shown in Table 5.3.
Table 5.3. Selected individual- and community-level interventions highlighted by the ECDC to address vaccine hesitancy
|
Target stakeholder |
Example interventions |
|---|---|
|
Individual-level interventions focusing on parents |
|
|
Individual-level interventions focusing on improving healthcare workers’ confidence and communication skills to respond to hesitant patients |
|
|
Community-level interventions |
|
Source: Adapted from ECDC (2017[115]), Catalogue of Interventions Addressing Vaccine Hesitancy, https://doi.org/10.2900/654210.
In addition to these interventions, behavioural approaches that aim to nudge the uptake of vaccines have recently gained prominence in improving vaccination uptake. For instance, one recent randomised controlled trial from Kenya demonstrated that in settings where the baseline vaccination coverage is already high, the use of mobile phone‑delivered reminders coupled with financial incentives can lead to important improvements in the uptake of vaccines for young children (Gibson et al., 2017[116]). Another field experiment from Sierra Leone that was implemented in the course of almost 2 years in 120 public clinics found that public health interventions that rely on principles grounded in behavioural economics like social signalling among community members can substantially increase vaccination uptake (Karing, 2018[117]).
Policies to tackle AMR outside the human health sector
Policies to tackle AMR in animal health
Globally, food consumption is rising faster than its production, exerting pressure on producers to resort to intensive, antibiotic-dependent agricultural practices in livestock and crop production. Many antibiotics used in food-producing animals are considered to be medically important for human medicine (Box 5.8). Plant production is another potentially important driver of antimicrobial use in agriculture, though relatively little is documented on the use of antibiotics in plants.
Box 5.8. WHO guidelines on the use of medically important antimicrobials in food-producing animals
Broadly, antibiotics are administered to animal populations for four main purposes:
Therapeutic use refers to instances when veterinary antimicrobials are administered in the presence of a clinically diagnosed infectious disease (WHO, 2017[118]).
Metaphylactic and prophylactic use occurs in the absence of a clinically diagnosed infectious disease. According to the WHO, metaphylactic use refers to treating animals that were exposed to animals with a clinical diagnosis of an infectious disease. Under these circumstances, antibiotic use is considered warranted, because the exposed animal may have been infected without presenting any symptoms (WHO, 2017[118]). In comparison, the WHO defined prophylactic use (i.e. disease prevention use) as the use of antimicrobials in healthy animals that are suspected to have a high risk of infection prior to the clinical onset of an infectious disease.
Growth promotion refers to antibiotic use in otherwise healthy animals to accelerate weight gain and improve the efficiency of feed utilisation (WHO, 2017[118]).
Recognising that many antimicrobials used in food-producing animals are either the same or closely related to those used in human medicine, in 2005, the WHO developed a systematic classification of medically important antimicrobials: important, highly important or critically important for human medicine (WHO, 2017[118]). These classifications provide the basis for the WHO List of Critically Important Antimicrobials for Human Medicines and guidance on veterinary antimicrobial use as follows (WHO, 2017[118]):
A complete restriction is recommended for the use of all medically important antimicrobials in food-producing animals for growth promotion purposes.
In the absence of disease, a complete restriction is recommended for the use of all classes of medically important antimicrobials in food-producing animals for the prevention of infectious diseases that have not yet been clinically diagnosed.
In the presence of disease, it is recommended that antimicrobials classified as critically important for human medicine should not be used to control the dissemination of a critically diagnosed infectious disease identified within a group of food-producing animals.
It is recommended that antimicrobials that are classified as the highest priority and critically important for human medicine should not be used for treating food-producing animals with clinically diagnosed infectious diseases.
Source: WHO (2017[118]), WHO Guidelines on Use of Medically Important Antimicrobials in Food-producing Animals, https://apps.who.int/iris/handle/10665/258970.
Over-reliance on veterinary antimicrobials can contribute to the growing AMR burden. Earlier reviews demonstrated the linkages between the use of veterinary antimicrobials and AMR in humans (Review on Antimicrobial Resistance, 2015[119]). One subsequent meta‑analysis quantified that reductions in veterinary antimicrobial use may contribute to a 10‑15% reduction in resistance in animals and a 24% reduction in humans (WHO, 2017[118]). In farm settings, contact with animals has been suggested to be a particularly important factor in AMR transmission (Hoelzer et al., 2017[120]), with evidence pointing to an increased risk of transmission of livestock-associated MRSA between animal species, from animals to humans (e.g. farmers in contact with animals) and to the environment (Klous et al., 2016[121]). In recognition of these complexities, one recent OECD publication examined the economic benefits and costs of the use of veterinary antimicrobials and provided five recommendations, three of which are particularly relevant for this publication that mainly focuses on human health aspects (Box 5.9). The rest of this section covers evidence associated with selected recommendations.
Box 5.9. OECD analysis of economic benefits and costs of antimicrobial use in food production animals
Recent OECD analysis sheds light on the economic benefits and costs associated with the use of veterinary antimicrobials in food-producing animals and provides five key recommendations (Ryan, 2019[122]). Three recommendations are particularly relevant for this publication that mainly focuses on human health:
1. Provide flexible regulations and a step-by-step approach to facilitate adjustment at the farm level. A regulatory approach that is likely to optimise prudent use may consist of various dimensions, including: i) quality veterinary services and clear legislation on the use of antimicrobials in animal production; and ii) good co‑operation and understanding of the regulations by all stakeholders supported by appropriate enforcement, adequate expertise and a well‑functioning surveillance system. Abolishing the use of veterinary antimicrobials for growth promotion should be prioritised.
2. Optimise a mix of management and biosecurity measures on the farm by: i) adopting good internal and external biosecurity; ii) increasing the natural immunity of the animals by improving breeding, housing, nutrition and stocking density on the farm; and iii) improving overall management on the farm.
3. Take an inter-sectoral or One Health approach to combat the negative externalities arising from AMR.
The two recommendations below are also crucial but relatively less evidence is available in terms of their effectiveness for human health:
1. Enhance information availability on economic benefits and costs of antibiotics in food-producing animals through: i) specific training and providing better information on the economic impact of antimicrobials to key stakeholders, especially farmers and veterinarians; ii) examples of “best practice” in terms of the optimal level of antimicrobial use on animal farms based on research in other countries and other animal enterprises; and iii) improvements in the diagnostic tests of animal diseases in order to optimise the use of preventive and affordable veterinary medication, as well as information on the antimicrobial classes best suited to treat and control the disease.
2. Improve the availability of information and knowledge on interventions alternative to antimicrobials and the relative costs and benefits associated with these interventions. It is also crucial to ensure adequate access to alternative interventions at affordable prices.
Source: Ryan, M. (2019[122]), “Evaluating the economic benefits and costs of antimicrobial use in food-producing animals”, https://doi.org/10.1787/f859f644-en.
Policies to promote prudent use of antimicrobials in animals

Policy interventions
Regulations concerning access to and use of veterinary antimicrobials.
Key messages
Regulations that restrict the use of veterinary antibiotics can result in reductions in AMR but the precise magnitude of the effectiveness of each type of regulation varies by setting.
Flexible regulations and a step-by-strategies that enable adjustments at the farm level are preferable to blanket bans that outlaw the use of antimicrobials for all purposes.
While considering regulatory options, priority should be given to regulations that limit antimicrobial use for growth promotion purposes.
The effectiveness of regulatory measures may be enhanced through the use of market mechanisms, voluntary initiatives, improving the availability of options alternative to antimicrobials and financial incentives for producers.
Regulations to optimise the prudent use of antimicrobials in animal populations
Regulations that seek to promote the prudent use of veterinary antimicrobials can be grouped in accordance with the degree to which they impose restrictions on access to and use of these antimicrobials. To date, some countries chose to outlaw the use of all veterinary antibiotics, while others imposed limits on a single antibiotic class or a single antibiotic for all indications of use (Tang et al., 2019[123]). Other countries restrict antimicrobial use for all non-therapeutic indications (e.g. limiting antimicrobial use for prophylaxis or growth promotion purposes). In recent years, some countries also started to incorporate multi-sectoral considerations in the rules and regulations around veterinary antimicrobials in line with the One Health framework (Box 5.10).
Box 5.10. Germany’s environmental checklist for the use of veterinary medicinal products
In 2017, the German Environment Agency published a checklist for veterinary medicine and animal husbandry, which delineates a list of environmental issues when using veterinary medicines (Umwelt Bundesamt, 2018[124]). A translation of these issues encompasses (OECD, 2019[125]):
Veterinary medicine
Carrying out sufficient levels of diagnostics and confirmation of a medical indication.
Possibility of avoiding a drug treatment, with consideration to input of pharmaceuticals to the environment, and to reducing resistance formation.
Informing the animal owner regarding the accurate administration of prescribed medicines, as well as correct storage and disposal.
Livestock farming
Considering the necessity of preventive measures, such as vaccinations.
Ensuring that alternative treatment methods are considered by veterinarians and farmers.
Complying with veterinary instructions for administrating the correct dosage, duration and frequency of medicine.
Avoiding unnecessary treatment and entry of pharmaceutical residues into the environment by keeping records of veterinary medicines.
Ensuring proper disposal of used medicines, leftovers, and packaging.
Ensuring a rest period of several months before the spreading of manure, which may contain traces of veterinary medicinal products.
Assessing what prevention measures could be taken at the farm scale to avoid or reduce future drug treatments.
Source: Umwelt Bundesamt (2018[124]), Umweltaspekte bei Verabreichung von Tierarzneimitteln, https://www.umweltbundesamt.de/umweltaspekte-bei-verabreichung-von#Landwirtschaft (accessed on 16 January 2021); OECD (2019[125]), Pharmaceutical Residues in Freshwater: Hazards and Policy Responses, https://doi.org/10.1787/c936f42d-en.
Most OECD countries have regulations in place that restrict access to veterinary antimicrobials (e.g. purchases only through authorised pharmacies, veterinarians and wholesalers and based on prescription). For instance, EU members started implementing new regulations in 2022 (i.e. Regulations (EU) 2019/6 and 2019/4), which outlawed the use of veterinary antimicrobials for prophylaxis purposes with certain exceptions and in medicated feed, and enforcing new restrictions concerning metaphylactic use. Moreover, the EU standards started covering imports from third parties outside the EU area (e.g. compliance with EU regulations that outlaw the use of veterinary antimicrobials as growth promoters). In comparison, in many LMICs, over-the‑counter purchase of veterinary antimicrobials without the need for a prescription remains the norm and access to veterinary antimicrobials is largely unchecked given the existing regulatory gaps and difficulties around enforcing existing regulations (Sulis et al., 2020[47]).
Available evidence suggests that regulating access to veterinary antimicrobials can result in reductions in AMR. One recent meta‑analysis quantified that a complete ban on antimicrobial use was associated with a 15% reduction in AMR (Tang et al., 2019[123]). However, a blanket ban on veterinary antimicrobials for all purposes may not be necessary. In their review, Tang et al. (2019[123]) suggested that regulations that allowed antimicrobial use for therapeutic purposes achieved similar levels of reduction in AMR, compared to regulations that outlawed all types of antimicrobial use. Among less restrictive regulations, those with narrower targets (e.g. targeting a specific antibiotic or an antibiotic class) were less effective than those that imposed broader limitations (Tang et al., 2019[123]). However, these findings should be interpreted with caution, as many of the studies that provide the basis for these results suffer from important methodological weaknesses. Nonetheless, in congruence with these findings, previous OECD analysis recommends flexible regulations and a step-by-strategies that enable adjustments at the farm level (Ryan, 2019[122]).
While considering regulatory options to optimise the prudent use of veterinary antimicrobials, limiting use for growth promotion purposes should be prioritised. This type of usage presents an alarming public health challenge because it entails exposing bacteria to antimicrobials in low doses over prolonged periods of time, which, in turn, elevated the risk of developing resistance. One meta‑analysis calculated that a 30% reduction in the proportion of antibiotic-resistant isolates may be achieved by restricting the use of antimicrobials for growth promotion, suggesting that such restrictions can offer a highly successful strategy for tackling AMR (Tang et al., 2019[123]).
Countries follow different regulatory paths while restricting the use of antimicrobials for growth promotion in their own setting. In 2017, the United States embarked on a regulatory process focusing on medically important antimicrobial drugs used in the feed or drinking water of food-producing animals (FDA, 2020[126]). With the introduction of new regulations, veterinary oversight has become a requirement for the purchase of medically important antimicrobial drugs and the use of these drugs’ growth promotion was outlawed (FDA, 2020[126]). China, another important agricultural producer, followed a different path in restricting antimicrobial use for growth promotion. In 2016, the country rolled out new restrictions, which entailed a ban on the use of Colistin for growth promotion – an antibiotic categorised as the highest priority and critically important for human medicine used as a last resort for treating multidrug-resistant Gram-negative infections (WHO, 2019[127]). These regulations were followed by new restrictions that were rolled out in 2019 that made it illegal to use medicated feed additives for growth promotion, except for traditional Chinese medicine (Hu and Cowling, 2020[128]).
While regulations are key to promoting the prudent use of veterinary antimicrobials, countries also make use of market mechanisms to limit the use of antimicrobials as growth promoters. Interestingly, one recent World Organisation for Animal Health (WOAH) survey revealed that 50 countries were able to enforce bans on antimicrobial use for growth promotion in their settings without an explicit regulatory framework (2020[129]). Instead, some of these countries restricted market access to these molecules altogether, while others relied on strategies like bans on the imports of selected molecules and scaling up monitoring of manufacturing companies to ensure antimicrobials were used only in veterinary medicine. These strategies were complemented with efforts that provided pig and poultry farmers alternatives to antimicrobials while highlighting the need for improved sanitation and hygiene practices in agricultural production (WOAH, 2020[129]).
In settings where regulatory frameworks are already in place to promote the prudent use of veterinary antimicrobials, voluntary initiatives can be considered to enhance the effectiveness of these regulations. For instance, in 2005, a public health programme from Quebec, Canada, promoted voluntary withdrawal of Ceftiofur consumption in hatcheries, a broad-spectrum, third-generation cephalosporin. This programme was associated with remarkable reductions in the prevalence of Ceftiofur resistance in Salmonella Heidelberg isolates from 2004 to 2006: from 62% to 7% in retail chicken and 36% to 8% in humans (Dutil et al., 2010[130]). Conversely, a brief re‑introduction of Ceftiofur in 2007 was associated with spikes in the prevalence of resistant bacteria in retail chicken and humans. In Japan, the voluntary withdrawal of off-label use of Ceftiofur in chicken hatcheries was linked to significant declines in resistance to broad-spectrum cephalosporin in E. coli in healthy broilers from 16.4% in 2010 to 4.6% in 2013 (Hiki et al., 2015[131]), suggesting that the beneficial effects of voluntary initiatives may be reaped in a relatively short period.
Improving the availability of interventions that are alternative to antimicrobials offers another important strategy to strengthen the effectiveness of regulatory approaches without exacerbating the burden of animal diseases. For instance, one randomised controlled trial from the states of Michigan and New York in the United States investigated the impact of antibiotic-free feeding practices in dairy calves, where farmers started using non-medicated milk replacers instead of those that contained broad-spectrum antibiotics like oxytetracycline and neomycin (Kaneene et al., 2008[132]). This switch towards antibiotic-free feeding was associated with increased susceptibility to tetracycline in Salmonella and Campylobacter spp. and E. coli in dairy calves over a 12‑month period (Kaneene et al., 2008[132]). Importantly, no measurable increases were observed in cattle diseases in the study period.
Additionally, offering financial incentives for producers in agricultural and aquaculture industries can help improve the effectiveness of regulatory approaches. The FAO estimates that, globally, 1.3 billion people rely on livestock production for their livelihood, which accounts for nearly 40% of total agricultural output in developed countries and about 20% in developing nations (FAO, 2020[133]). Interventions that restrict the use of veterinary antimicrobials may have economic implications that vary across industry types (Box 5.11). In recognition, one potentially successful strategy is to consider financial incentives that can help assuage financial concerns among producers over the potential loss of farm productivity.
Box 5.11. Health and economic impacts of the termination of growth promoters in Denmark
Denmark is among the pioneering countries that terminated the use of antimicrobials as growth promoters through a combination of regulatory and voluntary action. In 1995, Denmark discontinued the use of avoparcin for non-therapeutic purposes, which was followed by a similar limitation on virginiamycin use in broilers, cattle and finishing pigs in 1998 (FAO/DMOEF, 2019[134]). The same year, Danish producers volunteered to discontinue the use of all antimicrobials for growth promotion in finishing pigs. In 1999, the use of all antimicrobial growth promoters was outlawed for all food-producing animals. Today, veterinary antimicrobials can only be used for therapeutic or metaphylactic purposes, as prophylactic use is no longer permitted (FAO/DMOEF, 2019[134]).
Building on these efforts, Denmark introduced the Yellow Card scheme in 2010, with the aim of reducing the overall consumption of antimicrobials in pig production (Jensen and Hayes, 2014[135]). Rather than targeting all pig farmers, this intervention focuses on farmers that consume the highest levels of antimicrobials. At the outset, the programme identified a threshold for veterinary antimicrobial use (FAO/DMOEF, 2019[134]). Farmers that exceed this threshold receive a yellow card that is meant to serve as a warning. Upon receiving a yellow card, farmers and veterinarians work together to lower antibiotic consumption on the farm within a nine‑month time frame (FAO/DMOEF, 2019[134]). If a farmer fails to achieve the target reductions in antimicrobial consumption in this time frame, they receive a red card, which brings about mandatory reductions in the stocking density of animals on the farm (FAO/DMOEF, 2019[134]).
The Danish Ie underscores the complexities in regulating the use of antimicrobials in animals. Available evidence shows that the Danish efforts to terminate the use of antimicrobial growth promoters in the 1990s were associated with declines in the total volume of antimicrobials used in animals (Jensen and Hayes, 2014[135]). In the months following the initial ban on antimicrobial growth promoters, some increases in therapeutic use were observed in pig production but this surge was substantially lower than the overall reduction in the total volume of antibiotics used (FAO/DMOEF, 2019[134]). Similarly, available evidence suggests that the Yellow Card initiative was associated with declines in antimicrobial consumption, with the country achieving the target of a 10% decline in antimicrobial use for food-producing animals by 2013 (FAO/DMOEF, 2019[134]).
In terms of the economic impact of the termination of antimicrobial growth promoters, the overall losses in farm productivity varied by industry type. For instance, in pig production, productivity losses were estimated to average around EUR 1.04 per pig produced, which was partly driven by excess mortality and increased medication use (WHO, 2002[136]). This corresponds to approximately a 1% increase in overall production costs. In comparison, in poultry production, additional costs associated with the observed reductions in feed efficiency were almost entirely offset by savings from reduced antimicrobial use (WHO, 2002[136]).
Available evidence on the Yellow Card initiative suggests that there was a 1% decline in profits among farms that remained above the programme target threshold (FAO/DMOEF, 2019[134]). The decline in profits was largely attributable to the additional farm-level investments that were needed to lower antimicrobial use in line with the programme threshold (FAO/DMOEF, 2019[134]). These investments included additional expenses to cover animal vaccines, higher-quality feed and veterinary consultations, suggesting that the programme may have contributed to a shift in the emphasis on farming management practices that relied on antimicrobial use for disease prevention in these farms.
Source: FAO/DMOEF (2019[134]), “Tackling antimicrobial use and resistance in pig production: Lessons learned from Denmark”, Food and Agriculture Organization of the United Nations; Jensen, H. and D. Hayes (2014[135]), “Impact of Denmark’s ban on antimicrobials for growth promotion”, https://doi.org/10.1016/j.mib.2014.05.020; WHO (2002[136]), Impact of Antimicrobial Growth Promoter Termination in Denmark: The WHO International Review Panel’s Evaluation of the Termination of the Use of Antimicrobial Growth Promoters in Denmark, https://apps.who.int/iris/handle/10665/68357.
Policies to prevent the emergence and spread of infectious diseases in animals
Policy interventions
Optimising farm management and biosecurity measures
Increase the coverage of animal vaccines.
Key messages
Investing in farm management, biosecurity and animal vaccines contribute to reductions in the likelihood of the emergence and spread of resistant pathogens in farm settings.
Additional expenses incurred due to investing in farm management and biosecurity measures can be offset by savings achieved from reducing reliance on antibiotics.
Optimising farm management and biosecurity measures
The spread of infection among animals living in the same population may increase the likelihood of veterinary antimicrobial use while leading to reductions in farm productivity and increases in costs (Dewulf, 2019[137]). As discussed earlier in Chapter 4, an important strategy to prevent the spread of infection in animal populations relates to better farm management practices and enhanced external and internal biosecurity measures. Improving the existing farm management practices can boost the natural immunity in animal populations (e.g. improvements in breeding, housing, nutrition and stocking density on the farm) (Ryan, 2019[122]), while biosecurity measures can help reduce the likelihood of emergence and spread of pathogens environments (Alarcón, Alberto and Mateu, 2021[138]). In recent years, many OECD countries adopted holistic approaches to reducing the spread of infection among animals (Box 5.12).
Box 5.12. Sweden’s holistic approach to livestock production
Today, Sweden has the lowest use of antimicrobials in livestock production among the EU countries, thanks to a holistic approach that prioritises preventive health strategies, coupled with restrictions on the use of veterinary antimicrobials. As the first European country that banned the use of antimicrobial growth promoters in 1986, Sweden applies a national regulatory framework that sets the rules and guidelines for access to veterinary antibiotics. The Swedish Veterinary Association provides specific guidelines on antibiotic use, which emphasises the importance of preventive and biosecurity measures.
In general, antibiotics are considered a last resort and their use is meant to be supported by a number of factors including bacteriological diagnosis and susceptibility tests, food safety and environmental and economic concerns. Since 2013, a national list of antibiotics is being used, which clearly delineates antibiotics that can be used only in human medicine (FAO, 2020[139]). Access to antimicrobials is closely regulated, whereby the Medical Products Agency requires prescribed antibiotics to be purchased only from pharmacies and not from veterinarians, with some exceptions for acute treatment (FAO, 2020[139]).
A key component of the Swedish approach is close stakeholder collaboration. Veterinarians and farmers work closely to optimise animal health. Many farmers are assigned to specific veterinarians, who typically carry out either extension or clinical work. At the point of care, veterinarians examine animals and animal groups before antibiotics can be dispensed and these examinations are required to involve AMR risk assessments. Veterinarians also give advice on biosecurity measures, which are tailored to each farm. Farmers can also make use of several advisory tools for biosecurity (e.g. checklists for infection control on farms). In addition, administrative boards in counties are responsible for providing authorisation before any new animal housing can be constructed.
The Swedish approach underlines the importance of monitoring and surveillance of animal health. During routine farm visits, veterinarians collect data on the health status of herds, which provide the basis for herd-specific recommendations. Data collected during each visit are recorded in the Swedish Board of Agriculture database, which offers summaries on disease incidence and treatment within the herd (FAO, 2020[139]). Data collated from farms are then used for surveillance, benchmarking and goal setting. An additional database for post-mortem disease registration is also operational and it aids decisions regarding breeding practices and culling strategies. Since 2013, Sweden requires the monitoring of certain resistant bacteria, including S aureus, methicillin-resistant Staphylococcus pseudintermedius, other methicillin-resistant coagulase‑positive Staphylococci and carbapenemase‑producing Enterobacteriaceae (FAO, 2020[139]). When a laboratory suspects that samples collected by a veterinarian include any of these agents, the local country administrative board and the Swedish Board of Agriculture are notified.
Source: FAO (2020[139]), Tackling Antimicrobial Use and Resistance in Dairy Cattle, https://doi.org/10.4060/cb2201en.
Improvements in farm management and biosecurity measures reduce reliance on veterinary antimicrobials and AMR in animals. A recent study from 38 Japanese pig farms rolled out a strict application of an all-in and all-out system that disallowed the mixing of different animal groups (Isomura, Matsuda and Sugiura, 2018[140]). This intervention was associated with reductions in the circulation of pathogens, as well as reductions in the amount and variety of antibiotics used. Another study in Belgium rolled out a complex intervention in 61 Flemish pig herds, which combined new herd management methods, biosecurity measures (including animal vaccines) and anthelmintic therapy and recommendations for farmers on prudent use of animal antibiotics (Postma et al., 2016[141]). This intervention was associated with significant reductions in the use of antimicrobials considered to be critically important for human medicine, as well as a 52% reduction of AMR in pigs from birth to slaughter and 32% in breeding animals. In other settings, biosecurity measures yielded preventive returns for multiple diseases at the same time, while reducing the probability of transmission within the intervention sites (Manyi-Loh et al., 2018[142]).
A particular set of biosecurity measures that work well in one setting may not necessarily be suitable in others, underscoring the importance of collaboration across different stakeholders like veterinarians and farmers to optimise biosecurity strategies that best fit local needs. Previous OECD analysis suggested that carrying out biosecurity assessments in regular intervals may help decide which biosecurity strategies should be prioritised in a given setting while facilitating regular assessments of the changes in farm biosecurity over time and performance benchmarking (Ryan, 2019[122]).
An important debate around the implementation of farm management and biosecurity measures relates to concerns about costs and potential changes in farm productivity. Available evidence suggests that additional expenses may be offset by savings due to reductions in antimicrobial consumption. For instance, one recent study from Flemish pig herds showed that improvements in biosecurity measures were linked to increases in the number of weaned piglets per sow per year (+1.1), daily weight gain (+5.9 g/day) and reductions in mortality in the finisher period (−0.6%) (Postma et al., 2016[141]). Similarly, another study from 117 farrow-to-finish pig farms in Belgium found that biosecurity measures were cost-effective (Rojo-Gimeno et al., 2016[143]). This study found that costs associated with the rollout of new biosecurity measures (median +EUR 3.96/sow/year) were lower than the net cost reduction associated with reduced antibiotic use (median −EUR 7.68/sow/year). Importantly, cost reductions were driven primarily by the declines in antibiotic use for prophylactic treatment. This study further pointed to reductions in mortality in finishers (‑1.1%).
Animal vaccines
Similar to humans, increasing vaccination coverage in animal populations offers an important strategy to prevent infections and reduce the need for antibiotic use. A well-documented example comes from aquaculture practices in Norway, the world’s largest salmon producer. To tackle the rising burden of furunculosis, a bacterial fish disease that is present in wild salmon, Norway embarked on a large‑scale vaccination initiative in the late 1980s in the fish farming sector. This initiative was complemented with additional measures like zoning and spatial re‑arrangement of marine production sites to limit the spread of infections. Today, all domestic produce in Norway is vaccinated, with only about 0.03% of salmon estimated to receive at least one course of antimicrobial drugs (Midtlyng, Grave and Horsberg, 2011[144]). This corresponds to less than one tonne of antibiotics used per year. In comparison, Chile, the second largest salmon producer globally, uses about 300 tonnes of antibiotics each year (FAO, 2020[133]), though the Chilean salmon industry recently pledged a 50% reduction in veterinary antibiotic use by 2025.
The uptake and scale‑up of animal vaccines can reduce the need for antibiotic use without adversely impacting farm productivity. In Norway, efforts to scale up vaccination coverage in salmon populations coincided with the doubling of domestic salmon production between 2003 and 2014, suggesting that the reductions in antibiotic use were unlikely to hinder the expansion of production capacity (Midtlyng, Grave and Horsberg, 2011[144]). In Hungary, oral vaccination and medication against Lawsonia intracellularis (L intracellularis) for finishing pigs were associated with significant reductions in the prevalence of porcine proliferative enteropathy and concurrent improvements in average daily weight, a commonly used measure of farm productivity (Thaker, 2006[145]). Animal vaccines can also yield important gains even in settings where veterinary antibiotic use is already relatively low. For instance, in Denmark, vaccination against L intracellularis in pig herds led to a 79% reduction in the use of oxytetracycline (Bak and Rathkjen, 2009[146]). This vaccination effort was also associated with gains in average daily weight and carcass weight and reductions in the fattening period by eight days.
Policies to tackle AMR in plant health

Policy interventions
Regulations to promote the prudent use of antimicrobials in plant population.
Enhancing farm biosecurity measures and strengthening integrated pest management (IPM).
Key messages
Regulations to limit the use of antimicrobials that may impart resistance against critically important drugs for animal and human health in plant populations may help lower AMR transmission but important gaps exist in the existing regulatory arrangements across G7 countries, OECD members and key partners.
Mechanisms are lacking for monitoring pesticide use in plant production.
Improving farm biosecurity and strengthening integrated pest management approaches can help reduce the likelihood of the emergence and spread of diseases in plants.
Antimicrobials are used in plant populations to treat bacterial and fungal diseases, though important knowledge gaps need to be addressed to inform the development of effective AMR policies. Limited evidence suggests that antimicrobial use during plant production can potentially contribute to AMR by exerting selective pressure on pathogens in the environment, particularly those that thrive in the surrounding soil and water bodies (FAO/WOAH/WHO, 2019[147]). No systematically collected sources of data are available on a global scale to shed light on the volume of antimicrobials used as pesticides. While the dearth of data makes it difficult to quantify the precise magnitude of the challenge (Lomazzi et al., 2019[148]), the FAO estimates that, globally, the amount of antibiotics used in plant products constitutes around 0.2‑0.4% of total agricultural antibiotic consumption each year (FAO, 2020[133]).
Policies to promote prudent use of antimicrobials in plants
Regulatory frameworks for antimicrobial use in plant production
Limited data suggest that countries are diverging in their reliance on pesticides. Today, pesticide use in Argentina, China and the United States constitute around 70% of the global use, with China accounting for about half of global consumption (Pretty and Bharucha, 2015[149]). Over the last two decades, the use of pesticides in China grew fourfold, while it remained relatively stable in Germany and the United States. In addition, welcome reductions were observed in Denmark, France, Italy, Japan and the United Kingdom (Pretty and Bharucha, 2015[149]). Some of the observed reductions in country-level sales of pesticides across EU countries may be partly explained by the scale‑up of regulatory frameworks that relate to pesticide use in plant production, including the EU law on sustainable use of pesticides (Article 15 of Directive 2009/128/EC) (European Commission, 2009[150]), as well as increased use of IPM techniques, technological advancements in spray applications and formulations, and enhancements in disease forecasting models. As shown in, Figure 5.7, in 2020, the annual sales volume of fungicides and bactericides remained the highest in 2020 in France across those countries that report information to Eurostat (2022[151]).
Figure 5.7. Top 10 countries that report the highest annual sales volume of fungicides and bactericides to Eurostat, 2020
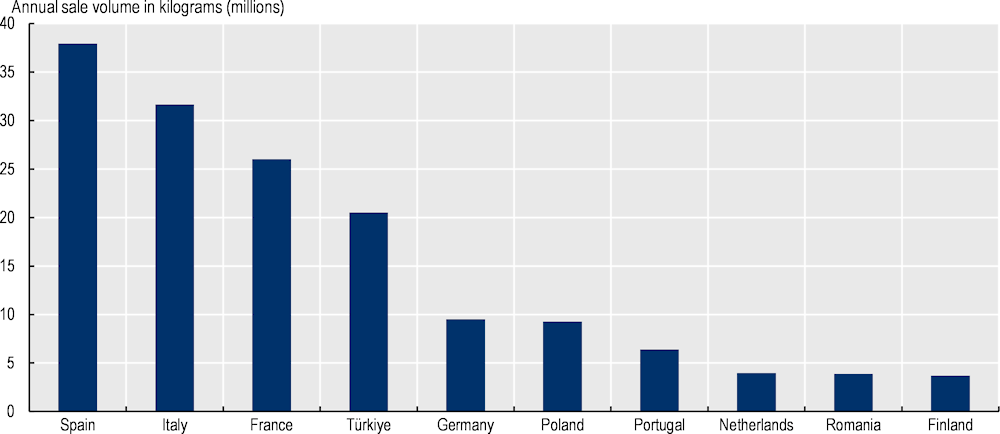
Source: Fungicide and bactericide sales data extracted from Eurostat (2022[151]), Pesticide Sales, https://ec.europa.eu/eurostat/databrowser/view/AEI_FM_SALPEST09/bookmark/table?lang=en&bookmarkId=53792fd3-191d-4201-aab5-c01c67fd927c (accessed on 26 October 2022).
Globally, there are important gaps in regulatory frameworks concerning antimicrobial use in plant production. In 2020‑21‑20, about 80% (124/155) of countries, globally, had some form of national policy or legislative framework in place to address quality safety and efficacy of pesticides, including antimicrobial pesticides (e.g. bactericides, fungicides), as well as their distribution, sale or use (WHO/FAO/WOAH, 2022[152]). The welcome news is that across G7 countries, OECD members and key partners, the availability of these regulatory frameworks were higher than the global averages, standing at around 90% (46/51). However, only 16% (8/51) of G7 countries, OECD members and key partners reported having enforcement and control mechanisms in place to ensure compliance with these frameworks in 2019‑20 (WHO/FAO/WOAH, 2022[152]).
In addition to gaps in regulatory frameworks, mechanisms are lacking monitoring of antimicrobial use in plant production. In 2020‑21, about 45% (23/51) of G20 countries, OECD members and key partners reported having some form of a national plan or mechanism in place to collect data and report the amount of pesticides sold/used at the national level, including antimicrobial pesticides, in order to respond to bacterial or fungal diseases (WHO/FAO/WOAH, 2022[152]). Relatedly, the existing guidelines concerning antimicrobial use in plants vary considerably across geographic regions. Globally, streptomycin is the most commonly recommended antibiotic for use in plants, followed by tetracycline and kasugamycin (Taylor and Reeder, 2020[153]). But notable geographic variations exist in current antibiotic guidance. For instance, tetracycline and streptomycin are most frequently recommended in the South-East Asia region, whereas producers in the Americas and Western Pacific regions rely more frequently on oxytetracycline and gentamicin (Taylor and Reeder, 2020[153]). This variation in antibiotic recommendations may be partly due to differences in prices, regulatory frameworks, product availability, cropping regimes, knowledge of agronomic advisors and the nature of pathogens that are of concern (Taylor and Reeder, 2020[153]).
Policies to prevent the emergence and spread of diseases in plants
Farm biosecurity, infection prevention and control, investing in IPM
It is crucial to implement AMR-relevant policies that can help prevent the emergence and spread of diseases in plant populations. Specifically, the FAO, WHO and WOAH guidance highlights the importance of enhancing farm biosecurity, and infection control measures (e.g. good hygiene practices and measures to prevent contamination) (FAO, 2020[154]), as well as the centrality of investing in IPM (FAO/WHO, 2019[155]). While the effects of these interventions in crop production on AMR transmission are largely unknown, they are expected to yield largely positive impacts for AMR by reducing the need for antimicrobial use in plant production.
The FAO further recommends the uptake and scale‑up of IPM approaches to supplement other efforts that aim to support healthy crop production. The FAO defines IPM as an approach to crop production and protection that brings together strategies to grow healthy crops (FAO, 2020[154]). By emphasising healthy crop production, IPM aims to minimise economic losses for crops while minimising risks for people and the environment caused by using pesticides (FAO/WHO, 2019[155]). Though limited, available evidence is in accordance with the FAO guidance. For instance, one recent study evaluated IPM systems across 24 countries in African and Asian regions over two decades and found that the IPM projects were associated with a nearly 41% increase in the yield of crops and an accompanying 31% decline in pesticide use (Pretty and Bharucha, 2015[149]).
Policies to tackle AMR in agri-food systems

Policy interventions
Building robust food safety compliance systems.
Scaling up integrated AMR surveillance.
Key messages
The Hazard Analysis and Critical Control Point (HACCP) system, a popular food safety compliance approach, can help reduce the burden of foodborne illness including food-related AMR, by supporting the focused implementation of food hygiene standards.
Evidence from OECD countries demonstrates that the introduction and robust enforcement of regulations that are aligned with international food hygiene and safety standards can help enhance the effectiveness of HACCP systems.
National AMR surveillance can help systematically monitor antimicrobial residues in foodstuffs.
Food plays an increasingly important role in the spread of AMR worldwide. It is estimated that almost 1 in 10 people globally fall ill after eating contaminated food, and roughly 420 000 deaths are attributed to foodborne diseases (FBDs) every year (Havelaar et al., 2015[156]). An important cause of concern is that the most common microbial agents causing FBD outbreaks are becoming drug-resistant, including norovirus, Campylobacter spp. and Enterobacteriaceae, a Gram-negative bacteria family composed of E. coli, Klebsiella, Salmonella or Shigella (Smith and Fratamico, 2016[157]). In recognition, the Codex Alimentarius Commission, the lead in setting reference food standards worldwide, sets out a wide range of standards, guidance and codes of practice that, collectively, aim to prevent the emergence of AMR and minimise its transmission in the food supply chain (FAO, n.d.[158]).
Food-producing animals and crops that carry resistant pathogens may eventually contaminate the food supply at any point in the farm-to-fork chain. In recognition, the AMR policies discussed earlier in relation to animal and plant health are instrumental in reducing AMR transmission in agricultural production. Supplementing these policies, it is crucial to build and scale up food safety compliance systems, which have been shown effective in interrupting AMR transmission in the food supply chain.
Scaling up food safety compliance systems
The introduction and scale‑up of food safety compliance systems is an important strategy to halt AMR transmission in the food supply chain. One commonly deployed preventive approach to food safety is the HACCP management system. The HACCP system entails identifying specific hazards associated with all stages of food production and implementing measures to address them to ensure food safety (FAO, 1997[159]). By intervening in all stages of food production, the HACCP system aims to prioritise the prevention of foodborne disease while reducing over-reliance on end-product testing (FAO, 1997[159]). It considers several inter-governmental codes of practice put forward by different international organisations, including the World Trade Organization (WTO), the WOAH and the Codex Alimentarius Commission.
Typically, the introduction of the HACCP system entails the application of seven essential principles pertaining to food handling including (FAO, 1997[159]):
1. Conduct a hazard analysis to identify the potential sources of hazards across all stages of food production.
2. Identify the critical control points and procedures to eliminate or minimise the likelihood of occurrence of hazards that have been identified.
3. Establish critical limits to ensure targets concerning critical control points are met.
4. Establish a monitoring system to track performance with respect to critical control points.
5. Establish corrective actions, which would be pursued if the critical control point targets are not met.
6. Establish verification procedures to ensure the HACCP system is working as intended (e.g. supplementary tests).
7. Establish record-keeping procedures to ensure proper documentation of the agreed procedures and their application.
In recent years, some OECD countries started incorporating the implementation of the HACCP system in their AMR national action plans (Box 5.13).
Box 5.13. Japan’s mandatory HACCP system in its food industry
In 2016, Japan reported a HACCP coverage rate of 30% in small- and medium-sized food business operators (FBOs), in comparison to the full coverage rate in France, the United Kingdom and the United States (MUFG, 2016[160]). The same year, the Japanese Ministry of Health, Labour and Welfare announced the nationwide introduction of the HACCP system covering the entire food chain adapted to Japan’s unique needs.
The introduction of Japan’s mandatory HACCP legislation came at a time when public concerns were rising over the risks of foodborne diseases (MHLW, 2016[161]). At this time, an analysis of the state of food safety in Japan identified that most cases of food contamination in the country could be attributed to the lack of proper implementation of good hygiene practices (e.g. maintenance and sanitation of establishments/equipment). In recognition, the Japanese Government proposed a new set of regulations that were more aligned with international food hygiene and safety standards (MHLW, 2016[161]). Today, this initiative is regarded as an important demonstration of Japan’s willingness to achieve high standards of food safety, as well as its inclination to expand its exports of foodstuffs.
A notable aspect of Japan’s food safety reform relates to the role of HACCP in Japan’s 2016‑20 National Action Plan on AMR. Unlike many other settings, Japan explicitly embedded the reinforcement of the implementation of HACCP in food processing and distribution establishments as a way to reduce occurrences of food poisoning and contamination of foodstuffs with antimicrobial-resistant organisms is explicitly embedded in its AMR mitigation strategy (Government of Japan, 2016[162]).
Source: MUFG (2016[160]), “The imminent mandatory implementation of HACCP in Japan”, https://www.murc.jp/english/report/quarterly_journal/qj1604_07/ (accessed on 21 January 2021); MHLW (2016[161]), Summary of the Final Report on the Implementation of Mandatory HACCP Program in Food Industry Adopted by the Ad Hoc Panel on International Standardization of Food Hygiene Control, https://www.mhlw.go.jp/english/topics/foodsafety/consideration/dl/summary_of_the_final_report.pdf; Government of Japan (2016[162]), National Action Plan on Antimicrobial Resistance (2016‑2020), https://www.mhlw.go.jp/file/06-Seisakujouhou-10900000-Kenkoukyoku/0000138942.pdf.
Though limited, available evidence suggests that HACCP systems are effective in interrupting AMR transmission in the food supply chain. Several earlier studies demonstrated that microbial contamination on kitchen surfaces and in food products was significantly reduced following the implementation of HACCP-based systems in food service establishments (Roy et al., 2016[163]; Soares et al., 2013[164]; Cenci-Goga et al., 2005[165]). Evidence emerging from other settings also shows similar improvements. For instance, one recent study showed that, in Thailand, slaughterhouses that implemented a HACCP food management system achieved reductions in Salmonella occurrence, as well as serotype numbers and serotype diversity (Wu et al., 2019[166]). Another study from the United States found that the publication of a pathogen reduction and HACCP rule in 1996 by the Food Safety and Inspection Service proceeded with an overall reduction in Salmonella occurrence on meat and poultry products between 2000 and 2018, though the study was unable to quantify the level of reduction in Salmonella occurrence attributable to the introduction of this rule (Williams et al., 2020[167]).
Scaling up integrated AMR surveillance
Integrated AMR surveillance can help systematically monitor antimicrobial residues in foodstuffs. Data generated through integrated surveillance can help facilitate the development and implementation of evidence‑based policies and inform decisions around resource allocation to activities that curb the spread of AMR through the food chain. These surveillance systems must collate data using clearly defined and harmonised methodologies that can facilitate the comparison of results not only within each country but also across countries.
There are several good practice examples from OECD countries that established integrated AMR surveillance systems including the food chain. For example, in Denmark, the Danish Integrated Antimicrobial Resistance Monitoring and Research Programme (DANMAP) also collects data not only from humans and animals but also from food products. Regional veterinary and food control authorities gather food samples from wholesale and retail outlets during their routine inspections for monitoring Salmonella and Campylobacter spp. or at the request of DANMAP to monitor enterococci and E. coli (Hammerum et al., 2007[168]). Importantly, information gathered through various resources are made available to the public through annual reports (DANMAP, 2020[169]). In the United States, the National Antimicrobial Resistance Monitoring System (NARMS) for enteric bacteria is the public health surveillance system that monitors AMR among enteric bacteria through data collected from humans, retail meats and food animals (CDC, 2022[170]) by multiple partners that play complementary roles, including the CDC, the U.S. Food and Drug Administration and the U.S. Department of Agriculture, among others. Specifically, each month, Salmonella, Campylobacter, Enterococcus and E. coli isolates are sent by health departments and universities in 19 states that participate in the sampling of retail meats to the Food and Drug Administration NARMS for serotyping, antimicrobial susceptibility testing and genetic analysis (CDC, 2022[170]).
Policies to tackle AMR in the environment
Policy interventions
Implementing measures to safely dispose of and remove antibiotics from the environment.
Upgrading municipal wastewater treatment facilities.
Improving wastewater management in pharmaceutical manufacturing facilities.
Improving waste management in agricultural production.
Improving waste collection and management in healthcare settings.
Encouraging proper pharmaceutical waste disposal in households.
Key messages
Upgrading technologies used in municipal wastewater treatment facilities can help reduce AMR transmission in the environment but none of these technologies can completely eliminate resistant bacteria and genes in their entirety.
In agricultural production, investing in integrated waste and manure management in the continuum of production can help reduce the likelihood of AMR transmission in the environment.
In healthcare settings, waste management, coupled with antimicrobial inventory control measures and environmental risk assessments offer a promising avenue for interrupting AMR transmission.
Drug take‑back programmes can help curb the inappropriate disposal of antimicrobials in households.
The environment can contribute to AMR through several pathways, though important gaps in knowledge exist. The environment can act as a transmission route for resistance by the way of water and soil systems (e.g. sewage systems, faecal contamination of water and pollutants that exert selective pressure for developing resistance) (Keen and Montforts, 2011[171]). The environment can also serve as a reservoir of resistant pathogens because many resistant genes have their origins in bacteria that live in the environment (Keen and Montforts, 2011[171]; Bengtsson-Palme, Kristiansson and Larsson, 2017[172]). In addition, horizontal gene transfers are another prominent resistance mechanism in the environment, particularly for most Gram-negative bacteria (Keen and Montforts, 2011[171]). However, relatively little is known about these biological processes, both in terms of transmission and evolution of resistance (Larsson et al., 2018[173]). Mirroring this, evidence is limited in terms of the effectiveness of technological, social, economic and behavioural interventions that can curb the emergence and spread of AMR through environmental channels (Larsson et al., 2018[173]).
The role of the environment as a contributing factor to AMR is increasingly acknowledged. In recent years, global and regional bodies are putting a spotlight on the role of environmental systems on AMR in line with the One Health approach. For instance, the 2017 United Nations Environmental Program (UNEP) Frontiers report noted that AMR is among the leading emerging issues of environmental concern with global implications (UNEP, 2017[174]). More recently, OECD analysis explored the role of pharmaceuticals, including antibiotics, in the environment and provided policy recommendations (Box 5.14). The rest of this section looks at available evidence in line with these recommendations.
Box 5.14. OECD Studies on Water Pharmaceutical Residues in Freshwater: Hazards and Policy Responses
Previous OECD analysis proposed four strategies to manage pharmaceuticals in the environment, including antimicrobials (OECD, 2019[125]). Specifically, the OECD analysis concluded that preventive measures that intervene early in a pharmaceutical’s life cycle may yield the greatest long-term societal return. Specifically, the OECD analysis highlights the importance of the following:
1. Improvements in monitoring and reporting on the occurrence, fate, toxicity, and human health and ecological risks of pharmaceutical residues in order to lay the ground for pollution reduction policies. This approach also entails considering the inclusion of environmental risks in the risk‑benefit analysis of the authorisation of new pharmaceuticals, and risk intervention and mitigation approach for pharmaceuticals with high environmental risk.
2. Implementing source‑directed approaches, such as the sustainable design and procurement of pharmaceuticals, to prevent the release of pharmaceutical residues into water bodies.
3. Introducing use‑orientated approaches, such as disease prevention, improved diagnostics and restrictions on pharmaceuticals with high environmental risk, to reduce inappropriate and excessive consumption of pharmaceuticals.
4. Implementing end-of-pipe measures, such as advanced wastewater treatment, public collection schemes for unused pharmaceuticals and education campaigns, to safely dispose of and remove pharmaceutical residues.
Source: OECD (2019[125]), Pharmaceutical Residues in Freshwater: Hazards and Policy Responses, https://doi.org/10.1787/c936f42d-en.
Upgrading municipal wastewater treatment plants
Wastewater is one prominent pathway through which antimicrobials are disseminated into the environment. Aquatic environments are amongst the most important reservoirs of antimicrobial-resistant agents and bacteria. Specifically, wastewater is a particular concern. Globally, only about 20% of wastewater that is directly discharged into the environment is treated (FAO, 2018[175]). Wastewater also plays an important role in agricultural production across the world, with at least one in ten people consuming food from plants that are irrigated with wastewater (WHO/WOAH/FAO, 2018[176]). Samples collected from wastewater from influent and effluent in municipal wastewater treatment plants (WWTPs), industrial and agricultural production sites and health facilities point to elevated levels of antibiotic-resistant bacteria (e.g. E. coli, Klebsiella spp., Shigella spp., Salmonella spp., Vibrio spp., Acinetobacter spp. and Enterococcus spp.) and genes (Fouz et al., 2020[177]). The final effluent discharged into the environment from these sites can contaminate the receiving water bodies if antimicrobial-resistant agents and bacteria are not removed completely (OECD, 2019[125]).
Upgrading WWTPs can help interrupt the transmission of AMR in the environment but available technologies offer different levels of removal efficiencies (OECD, 2019[125]; Shekhawat, Kulshreshtha and Gupta, 2020[178]). For instance, one study from England (United Kingdom) collected data from 20 WWTPs and found that terminal ultraviolet (UV) light treatment technology was the most effective option to reduce the levels of E. coli, while secondary and tertiary treatment yielded lower levels of reduction (Raven et al., 2019[179]). Importantly, this study concluded that even the most stringent treatment options such as tertiary treatment including UV light fell short of eradicating extended-spectrum B-lactamase producing E. coli (ESBL‑EC) from most wastewater effluent samples. Another study from 16 urban WWTPs in 10 European countries found that lower levels of antibiotic-resistant genes were released into the environment in WWTPs that were equipped with secondary clarifiers (Cacace et al., 2019[180]).
WTTPs provide a readily accessible avenue for AMR surveillance and monitoring. With the advent of new sequencing technologies, samples from WWTP outflows have been suggested as another avenue for monitoring the detection of new and circulating antibiotic resistance genes (Raven et al., 2019[179]; Larsson et al., 2018[173]). For instance, one study used metagenomic analyses and ribosomal ribonucleic acid (rRNA) sequencing on samples from 32 WWTPs influents in 17 major Chinese cities and detected 381 different genes that were resistant to almost all antibiotics (Su et al., 2017[181]). Importantly, these genes were shared extensively across cities with no apparent geographic clustering. Another study in Norway deployed whole‑genome sequencing to demonstrate that the same ESBL-EC type was shared in recreational waters, wastewater in close proximity to a WWTP and urine samples collected from humans residing in the same area (Jørgensen et al., 2017[182]).
Wastewater management in pharmaceutical manufacturing facilities
In countries with large antibiotic production capacity, a particular concern for AMR transmission relates to the release of the compounds generated during the manufacturing process into the environment. One well‑documented example comes from India – one of the world’s leading manufacturers of antibiotics. In the city of Hyderabad, one of India’s most populous cities, alarmingly high levels of ciprofloxacin were detected in the effluent samples collected from a WWTP that served 90 bulk pharmaceutical manufacturers (Larsson, de Pedro and Paxeus, 2007[183]). Notably, all of the bacteria detected in this WWTP were multidrug resistant and high levels of resistant genes were detected in the surface water in downstream rivers up to 17 km from the WWTP site (Kristiansson et al., 2011[184]). Samples from the groundwater and drinking water in nearby villages were shown to contain a variety of pharmaceuticals (Fick et al., 2009[185]). In response, the Indian Government announced in 2020 a new bill that consisted of restrictions on the level of residue from 121 common antibiotics that can be disseminated into the environment from pharmaceutical manufacturers (EPR, 2020[186]).
Even in settings where regulatory frameworks are in place, wastewater management in pharmaceutical manufacturing sites remains a concern. For instance, one recent study in the United States collected samples from 20 WWTPs and found that the concentration of 33 pharmaceuticals was significantly greater in WWTPs linked to pharmaceutical production sites, compared to those that were not connected to similar facilities (Scott et al., 2018[187]). In Europe, the European Medicines Agency considers environmental risk assessment of new pharmaceuticals before the required authorisations can be complicated before entry into the market (OECD, 2019[125]). Yet, antibiotic-resistant microorganisms have been observed in European water bodies that receive a discharge from pharmaceutical production sites (Nappier et al., 2020[188]).
In the face of these challenges, it is paramount to promote co‑operation and collaboration across different stakeholders to develop industry standards for the management of waste/wastewater in manufacturing facilities and to achieve high rates of compliance among manufacturers. To this end, in 2020, the WHO published practical guidelines for pharmaceutical manufacturers, inspectors and national regulatory bodies for handling antimicrobial waste and/or process effluents from pharmaceutical processes (WHO, 2020[189]). Concurrently, many pharmaceutical companies are making efforts to develop and implement industry standards for the environmental management of the manufacturing process of antibiotics (Box 5.15).
Box 5.15. AMR Industry Alliance framework for assessing the environmental impact of manufacturing
In recent years, a number of private sector coalitions proliferated to scale up efforts to tackle AMR in manufacturing sites. One of these coalitions is the AMR Industry Alliance, a broad consortium of companies and associations. Members of the AMR Industry Alliance are estimated to represent about one‑third of the global antibiotic supply and almost half of the AMR-relevant products currently under clinical development. The AMR Industry Alliance commits its members to an industry roadmap (Tell et al., 2019[190]), which promotes:
Reducing the environmental impact of the production of antibiotics.
Helping ensure antibiotics are used only by patients who need them.
Improving access to current and future antibiotics, vaccines and diagnostics.
Exploring new opportunities for open collaborations between industry and the public sector.
In 2018, the members of the AMR Industry Alliance published a framework for managing antibiotic discharge in manufacturing sites, with the aim of minimising the environmental impact of manufacturing. This framework delineates minimum requirements to carry out risk evaluation, lays out a methodology for evaluating risks as well as risk-based targets for antibiotic discharge from manufacturing sites of about 120 antibiotics (AMR Industry Alliance, 2020[191]). These targets are meant to be used by all antibiotic manufacturing companies, as they establish their own targets for antibiotic residue in wastewater from their plants. By 2020, about 80% of AMR Industry Alliance members have reportedly assessed risks in their manufacturing sites against the new industry targets and 82% indicated that they either met or partially met the minimum requirements (AMR Industry Alliance, 2020[191]).
Moving forward, the most recent progress report of the AMR Industry Alliance makes recommendations towards accelerating efforts to make surveillance data publicly available, which includes information on infection rates, AMR patterns and antibiotic use, supports local health system and laboratory capabilities for the diagnosis and treatment of drug-resistant infections and fosters partnerships with other stakeholders (AMR Industry Alliance, 2020[191]).
Source: Tell, J. et al. (2019[190]), “Science‐based targets for antibiotics in receiving waters from pharmaceutical manufacturing operations”, https://doi.org/10.1002/ieam.414; AMR Industry Alliance (2020[191]), 2020 Progress Report, https://www.amrindustryalliance.org/wp-content/uploads/2020/01/AMR-2020-Progress-Report.pdf.
Waste management in agricultural production
Agricultural production has important consequences for AMR transmission in the environment, with impacts in magnitudes comparable to WWTPs and healthcare facilities. In farm settings, veterinary antibiotics are disposed into the environment through animal waste, animal excrete re‑used as manure and runoff from animal waste storage and disposal tanks (FAO, 2018[175]; Nappier et al., 2020[188]; Hoelzer et al., 2017[120]). Additionally, wastewater from livestock production is widely utilised in the form of organic fertilisers and soil conditioners. These pathways through which veterinary antibiotics enter the environment have important implications for AMR transmission. For instance, one study that collected samples from wastewater across 96 countries showed that the abundance of antimicrobial-resistant genes from swine and poultry farms was three to five times that of the magnitude observed in hospital and municipal wastewater (He et al., 2020[192]). This study also showed that wastewater samples collected from cattle and fish farms had similar levels of antimicrobial-resistant genes compared to those collected from hospitals and WWTPs.
In recognition, several livestock waste treatment technologies can be considered to mitigate the role of livestock waste in environmental contamination, including anaerobic digestion, thermophilic or mesophilic composting, biological treatment process and constructed wetlands (He et al., 2020[192]). Similar to the technologies used in WWTPs, each livestock waste treatment technology comes with a different set of advantages and caveats, and their efficiency may depend on the local operating conditions and manure type. Importantly, none of them guarantees that antibiotic-resistant bacteria and genes are eliminated from livestock waste in their entirety.
Importantly, investments in waste management technologies should be made in accordance with local needs and conditions. In their multi-country study, He et al. (2020[192]) suggested that the level of abundance may be correlated with the intensity with which antibiotics are used in agricultural production, as well as the resulting concentration of residual antibiotics. For instance, this study showed that the highest absolute abundance of antimicrobial-resistant bacteria was observed in samples collected from livestock waste in China, the largest producer and consumer of antibiotics in the world. However, this study found that country-level aggregate data on antibiotic use may mask important realities at the farm-level antibiotic use. For instance, He et al. (2020[192]) also compared tet in swine wastewater in Shandong, China, and the state of Colorado in the United States, two countries with comparable levels of country-aggregated antibiotic use. This study concluded that the abundances of antimicrobial-resistant genes were higher in Shandong than in Colorado, suggesting that it is important to keep in mind the site‑specific chemical and physical conditions that contribute to differences in the growth of antimicrobial-resistant bacteria and the propagation and attenuation of antimicrobial-resistant genes.
The WHO, WOAH and FAO promote integrated manure management practices in the continuum of manure use, from collection to storage and treatment before re‑application as fertiliser or disposal. In many farms, animal excrete is re‑used as manure as a way to support food and feed production in farms (WHO/WOAH/FAO, 2018[176]). However, the application of manure is associated with significant increases in the diversity and abundance of antimicrobial-resistant genes in the soil, with abundance in manured soil is estimated to reach up to 28 000 times that of the abundance in un-manured soil (He et al., 2020[192]). While the contribution of manure to total fertiliser use has been on a decline, manure remains a key input into agricultural production in many LMICs, particularly in Africa (84%) and Latin America (73%) regions (FAO, 2018[175]). In recognition, the WHO, WOAH and FAO provide guidance for countries to aid efforts to optimise manure management practices in their own settings by improving the ways in which manure is stored, treated, handled and disposed of (WHO/WOAH/FAO, 2018[176]).
Waste collection and management in healthcare settings
According to WHO estimates, about 15% of healthcare waste is considered hazardous, which may contribute to spreading drug-resistant microorganisms in the environment (WHO, 2017[193]). Despite this, globally, 40% of health facilities lack systems that can ensure the safe disposal of healthcare waste where antibiotic-resistant microorganisms may be present (WHO/WOAH/FAO, 2018[176]).
Even so, available evidence on the role of healthcare settings in AMR transmission in the environment remains mixed. To date, elevated levels of antimicrobial-resistant agents and genes have been reported in hospital effluent wastewater samples and water bodies receiving untreated hospital waste (Hocquet, Muller and Bertrand, 2016[194]; Fouz et al., 2020[177]). In contrast, one recent study from ten European countries found that effluent from hospitals represented only around 0.2‑2% of the total wastewater in urban settings and that the number of hospitals and hospitalised patients did not correlate with the amount of antibiotic-resistant genes released from WWTPs in urban areas (Cacace et al., 2019[180]). Similarly, another study from 20 WWTPs across the East of England (United Kingdom) region showed that there were no statistically significant differences in ESBL-EC counts between samples taken from WWTPs that directly received waste from acute care hospitals and those that did not (Raven et al., 2019[179]). Taken together, these findings suggest that the role of healthcare facilities in AMR transmission into the environment may be closely related to the unique circumstances in each setting.
The selection of optimal wastewater management strategies in healthcare settings depends largely on whether these facilities are connected to a WWTP system. The WHO recommends that in cases where antimicrobial waste from a healthcare facility goes directly into a WWTP, decentralised wastewater treatment at that health facility may not be necessary (WHO/WOAH/FAO, 2018[176]). In line with this recommendation, one study from Switzerland showed that the introduction of early separation and onsite treatment of wastewater in hospitals led to lower levels of emission of contaminants of emerging concern to the environment, including pharmaceuticals (EAWAG, 2007[195]). However, this study also found that the annual operational cost of this decentralised wastewater system was considered high and that this strategy fell short of offering a more cost-effective option than upgrading centralised municipal WWTP. In comparison, the WHO recommends that if wastewater from a health facility does not go to a central WWTP, then pre‑treatment at the health facility is needed to ensure reductions in pathogens and AMR (WHO/WOAH/FAO, 2018[176]). In these cases, the WHO indicates that the choice of wastewater treatment technology should optimise for minimising AMR release and not rely on conventional waste treatment technologies.
Efforts to optimise waste management in healthcare settings can be bolstered by antimicrobial inventory control measures. The WHO recommends that antimicrobial waste should be kept separate from other waste, encapsulated, buried, incinerated or returned to manufacturers (WHO/WOAH/FAO, 2018[176]). Waste minimisation techniques can also be considered, whereby inventories for high-use pharmaceuticals, including antimicrobials, can be maintained, and antimicrobials with short expiration dates may be redistributed to other health facilities in the area where there may be a need. In recent years, OECD countries are introducing novel approaches to integrating environmental considerations into other aspects of efforts to minimise the role of the health sector in the AMR burden (Box 5.16).
Box 5.16. Integrating environmental considerations to the promote prudent use of antibiotics in Sweden
In 1996, Sweden introduced a new regulation that stipulated that each healthcare region in the country should establish at least one Drug and Therapeutics Committee that is responsible for developing guidelines and recommendations that promote the prudent use of antibiotics. In 2000, the Stockholm Healthcare Region published the “Wise List” which provides recommendations for the use of 212 essential medicines that are used across different levels of care in the public and private sectors including primary care and hospitals spanning 24 therapeutic areas (Regional Drug Expert Consortium, 2011[196]). The list also includes non-pharmacological advice for certain therapeutic areas and recommendations tailored to reflect case severity and comorbidities. Starting from 2008, adherence to Wise List recommendations was linked to provider payment for prescribers in primary care (Eriksen et al., 2017[197]).
A novel aspect of the Wise List is that it classifies pharmaceuticals in line with their potential impact on the environment. This approach explicitly recognises that pharmaceuticals can remain in the environment long after their initial use, especially in water resources. In doing so, Wise List recommendations aim at minimising environmental risks attributable to the use of pharmaceuticals in healthcare settings. For each pharmaceutical substance, the environmental risks are examined both in terms: i) environmental hazard, defined as the capacity to cause damage to the environment (i.e. persistence, bioaccumulation and toxicity); and ii) environmental risks, referred to as the risks of toxicity to the aquatic environment (Eriksen et al., 2017[197]).
Evidence suggests that adherence to Wise List recommendations for prescribers in the Stockholm Healthcare Region increased from 75% to 84% between 2000 and 2015, with variation across different practice types (e.g. primary care versus hospitals) narrowing over time (Eriksen et al., 2017[197]). One explanation for this finding relates to the highly transparent process through which Wise List recommendations have been developed. This process involved input from various stakeholders including clinicians, clinical pharmacologists, pharmacists and nurses based on a review of existing scientific evidence (Eriksen et al., 2017[197]). Compounding the beneficial impact of this process was the use of evidence‑based guidelines in combination with an effective communication strategy and continuous medical education opportunities.
Source: Regional Drug Expert Consortium (2011[196]), “The ‘Wise List’- A comprehensive concept to select, communicate and achieve adherence to recommendations of essential drugs in ambulatory care in Stockholm”, https://doi.org/10.1111/j.1742-7843.2011.00682.x; Eriksen, J. et al. (2017[197]), “High adherence to the ‘Wise List’ treatment recommendations in Stockholm: A 15‑year retrospective review of a multifaceted approach promoting rational use of medicines”, https://doi.org/10.1136/bmjopen-2016-014345.
Pharmaceutical waste disposal in households
Inappropriate disposal of antimicrobials in households remains an important pathway through which antibiotics are disseminated into the environment. While quantifying the precise magnitude of this challenge is difficult, recent estimates suggest that about 10‑50% of medicines are disposed of by households through sinks and bathrooms (OECD, 2019[125]). When not properly discarded or eliminated during wastewater treatment, antibiotics can enter aquatic environments and promote resistance even in small doses (Tong, Peake and Braund, 2011[198]).
In response, many OECD countries are rolling out drug take‑back programmes but the evidence is lacking in terms of their effectiveness for curbing AMR. For instance, all EU members are obligated to implement medicine collection and disposal schemes for unused medicines in their countries (HCWH-Europe, 2013[199]). But the ways in which drug take‑back programmes are implemented in each EU country vary. In Sweden, the Swedish Pharmaceutical Society spearheads a government-funded, national programme for the safe disposal and destruction of unused medications, in conjunction with wholesalers and community pharmacies (Persson, Sabelström and Gunnarsson, 2009[200]). In other countries like Belgium, Canada, France and Spain, collection schemes are funded in accordance with the Extended Producer Responsibility principle, whereby pharmaceutical companies are required to collect and destroy unused medicines that they produce (OECD, 2019[125]). The scope of medicines covered by these schemes also differs across countries. While most drug take‑back programmes focus only on pharmaceuticals used in human medicine, others, like Portugal’s national collection system, have extended their scope to include veterinary medicines (HCWH-Europe, 2013[199]).
Emerging lessons
This chapter presented an overview of the current state of evidence on AMR in line with the One Health framework. Looking across countries and policies to address AMR, several important lessons emerge:
In human and animal health, flexible AMR policies that aim to create an enabling work environment that promotes prudent use of antimicrobials achieve similar levels of improvements in antibiotic prescribing behaviours compared to restrictive policies that limit the opportunities for using antibiotics. In human health, the WHO guidance indicates that restrictive interventions that limit the opportunities for antimicrobial prescriptions may result in relatively quick gains in prudent antibiotic prescribing behaviours. But, over time, these interventions achieve similar levels of improvement in prescribing behaviours compared to interventions that deploy persuasive strategies (e.g. education and feedback interventions) that aim to induce behavioural change by enabling a professional environment that promotes prudent prescribing behaviours (WHO, 2019[20]). Importantly, supporting restrictive interventions with persuasive ones was shown to improve the effectiveness of restrictive policies. In animal health, available evidence suggests that regulations that allow the use of antibiotics for therapeutic purposes may be just as effective as bans on all types of antibiotics used in food-producing animals (Tang et al., 2019[123]). Similarly, limitations on antimicrobial growth promoters should be prioritised, as these restrictions have been shown to yield substantial reductions in AMR burden. This is in line with the previous OECD analysis that recommends flexible regulations and stepwise adjustments at the farm level.
Getting input and buy-in from key stakeholders (e.g. healthcare providers, veterinarians, farmers) in the design and implementation of AMR policies improves the effectiveness of these policies. The process of designing and implementing AMR policies entails interactions across stakeholders from multiple sectors with diverse interests, influences and positions towards each policy option (e.g. healthcare providers, veterinarians, farmers and pharmaceutical manufacturers, local and national regulatory bodies). Getting buy-in from these stakeholders can help improve the likelihood that the policy objectives will be achieved. In human health, the chapter found evidence that embedding provider feedback interventions in persuasive ASP strategies yielded greater compliance with ASP guidelines. In animal health, the chapter highlighted evidence from Denmark and Sweden, which demonstrated that co‑operation across farmers, veterinarians and local and national regulatory bodies were paramount to successfully eradicating the use of antimicrobial growth promoters. Concerning AMR transmission in the environment, the chapter showed that the involvement of pharmaceutical manufacturers has played an important role in supporting national and international efforts to develop industry standards to curtail the adverse effects of industrial production on AMR transmission.
The effectiveness of AMR policies may change over time, necessitating modifications in the design and implementation of these policies to address evolving needs in a given setting. The chapter showed that, in countries with long-standing vaccination programmes, the beneficial effects of high rates of vaccination coverage have been shown to accrue over time. Conversely, the chapter found evidence that the removal of an effective AMR intervention may threaten gains achieved over time. This was the case in Canada where a temporary re‑introduction of Ceftiofus in 2007 was followed by a rise in the prevalence of resistant bacteria in animals and humans. Relatedly, even when an intervention is effective at the outset, the beneficial effects may be attenuated over time. These changes in the effectiveness of AMR policies highlight the importance of tracking the performance of these policies over time and introducing modifications in their design and implementation to reflect the evolving health needs in a given context.
Even when AMR interventions are effective, unintended consequences may occur. For instance, in the United Kingdom, the rollout of a mobile application that aimed to improve access to antibiotic guidelines was associated with improvements in the fraction of antibiotic prescriptions in compliance with the existing guidelines, while also leading to declines in the completeness of documentation of prescriptions (Charani et al., 2017[32]). In South Korea, the launch of a national programme that separated dispensing from prescribing of antimicrobials led to improvements in the appropriate prescription of antibiotics but this programme also prompted a rise in medical expenditures, partly due to increases in provider fees (Park et al., 2005[64]). Taken together, these findings suggest that it may be beneficial to consider the potential effects of AMR policies before they are rolled out – including potential effects that may not necessarily be the main intent of the intervention – and attempt to pre‑empt negative consequences.
Improving the availability and accessibility of information from novel data sources offers an important avenue for supporting rigorous evaluations of AMR policies in line with the One Health framework. Welcome news is that the analytical basis that evaluates the impact of AMR interventions has grown since the previous OECD analysis. Despite this, many systematic reviews and meta‑analyses underline the urgent need for higher-quality studies. One important factor that hinders rigorous policy evaluations relates to the dearth of systematically collected data. In recognition, data collected from global AMR surveillance networks (e.g. the Global Antimicrobial Resistance and Use Surveillance System) offer a valuable avenue for systematic data collection. In addition, other novel data sources can be considered, including: data collated from HAI surveillance networks; data gathered through computerised decision support systems and e‑prescribing tools; data collected during veterinary visits to farms; and samples collected from WWTPs. Combined, these data sources can help support efforts to design and implement effective policies to stem the AMR tide.
References
[44] ACSQHC (2021), Antimicrobial Prescribing Practice in Australian Hospitals: Results of the 2019 Hospital National Antimicrobial Prescribing Survey, Australian Commission on Safety and Quality in Health Care, https://www.safetyandquality.gov.au/sites/default/files/2021-02/report_-_2019_hospital_naps.pdf.
[138] Alarcón, L., A. Alberto and E. Mateu (2021), “Biosecurity in pig farms: A review”, Porcine Health Management, Vol. 7/1, https://doi.org/10.1186/s40813-020-00181-z.
[93] Allen, G. et al. (2014), “A multitiered strategy of simulation training, kit consolidation, and electronic documentation is associated with a reduction in central line-associated bloodstream infections”, American Journal of Infection Control, Vol. 42/6, pp. 643-648, https://doi.org/10.1016/j.ajic.2014.02.014.
[48] Ammenwerth, E. et al. (2008), “The effect of electronic prescribing on medication errors and adverse drug events: A systematic review”, Journal of the American Medical Informatics Association, Vol. 15/5, pp. 585-600, https://doi.org/10.1197/jamia.m2667.
[191] AMR Industry Alliance (2020), 2020 Progress Report, https://www.amrindustryalliance.org/wp-content/uploads/2020/01/AMR-2020-Progress-Report.pdf.
[98] Andersen, B. et al. (2002), “Spread of methicillin-resistant Staphylococcus aureus in a neonatal intensive unit associated with understaffing, overcrowding and mixing of patients”, Journal of Hospital Infection, Vol. 50/1, pp. 18-24, https://doi.org/10.1053/jhin.2001.1128.
[137] Anderson, M. (ed.) (2019), Tackling antimicrobial resistance in the food and livestock sector, Cambridge University Press, https://doi.org/10.1017/9781108864121.
[90] Armellino, D. et al. (2011), “Using high-technology to enforce low-technology safety measures: The use of third-party remote video auditing and real-time feedback in healthcare”, Clinical Infectious Diseases, Vol. 54/1, pp. 1-7, https://doi.org/10.1093/cid/cir773.
[30] Arshad, M. (ed.) (2019), “Impact of a smartphone app on prescriber adherence to antibiotic guidelines in adult patients with community acquired pneumonia or urinary tract infections”, PLoS ONE, Vol. 14/1, p. e0211157, https://doi.org/10.1371/journal.pone.0211157.
[108] Atkins, K. et al. (2018), “Use of mathematical modelling to assess the impact of vaccines on antibiotic resistance”, The Lancet Infectious Diseases, Vol. 18/6, pp. e204-e213, https://doi.org/10.1016/s1473-3099(17)30478-4.
[26] Aziz, R. (ed.) (2017), “The effectiveness of computerised decision support on antibiotic use in hospitals: A systematic review”, Plos ONE, Vol. 12/8, p. e0183062, https://doi.org/10.1371/journal.pone.0183062.
[70] Bakhit, M. et al. (2019), “Exploring patients’ understanding of antibiotic resistance and how this may influence attitudes towards antibiotic use for acute respiratory infections: A qualitative study in Australian general practice”, BMJ Open, Vol. 9/3, p. e026735, https://doi.org/10.1136/bmjopen-2018-026735.
[146] Bak, H. and P. Rathkjen (2009), “Reduced use of antimicrobials after vaccination of pigs against porcine proliferative enteropathy in a Danish SPF herd”, Acta Veterinaria Scandinavica, Vol. 51/1, https://doi.org/10.1186/1751-0147-51-1.
[172] Bengtsson-Palme, J., E. Kristiansson and D. Larsson (2017), “Environmental factors influencing the development and spread of antibiotic resistance”, FEMS Microbiology Reviews, Vol. 42/1, https://doi.org/10.1093/femsre/fux053.
[81] Bianco, A. et al. (2018), “Prospective surveillance of healthcare-associated infections and patterns of antimicrobial resistance of pathogens in an Italian intensive care unit”, Antimicrobial Resistance & Infection Control, Vol. 7/1, https://doi.org/10.1186/s13756-018-0337-x.
[107] Bloom, D. et al. (2017), “Moving beyond traditional valuation of vaccination: Needs and opportunities”, Vaccine, Vol. 35, pp. A29-A35, https://doi.org/10.1016/j.vaccine.2016.12.001.
[13] Bloom, D. et al. (2021), “How new models of vaccine development for COVID-19 have helped address an epic public health crisis”, Health Affairs, https://doi.org/10.1377/hlthaff.2020.02012.
[106] Buchy, P. et al. (2020), “Impact of vaccines on antimicrobial resistance”, International Journal of Infectious Diseases, Vol. 90, pp. 188-196, https://doi.org/10.1016/j.ijid.2019.10.005.
[180] Cacace, D. et al. (2019), “Antibiotic resistance genes in treated wastewater and in the receiving water bodies: A pan-European survey of urban settings”, Water Research, Vol. 162, pp. 320-330, https://doi.org/10.1016/j.watres.2019.06.039.
[170] CDC (2022), About NARMS: Tracking Trends in Resistance, Centers for Disease Control and Prevention, https://www.cdc.gov/narms/about/index.html (accessed on 21 June 2022).
[165] Cenci-Goga, B. et al. (2005), “Effect of the implementation of HACCP on the microbiological quality of meals at a university restaurant”, Foodborne Pathogens and Disease, Vol. 2/2, pp. 138-145, https://doi.org/10.1089/fpd.2005.2.138.
[32] Charani, E. et al. (2017), “Effect of adding a mobile health intervention to a multimodal antimicrobial stewardship programme across three teaching hospitals: an interrupted time series study”, Journal of Antimicrobial Chemotherapy, Vol. 72/6, pp. 1825-1831, https://doi.org/10.1093/jac/dkx040.
[86] Charrier, L. et al. (2008), “Integrated audit as a means to implement unit protocols: A randomized and controlled study”, Journal of Evaluation in Clinical Practice, Vol. 14/5, pp. 847-853, https://doi.org/10.1111/j.1365-2753.2008.01042.x.
[12] Chou, R. et al. (2020), “Epidemiology of and risk factors for coronavirus infection in health care workers”, Annals of Internal Medicine, Vol. 173/2, pp. 120-136, https://doi.org/10.7326/m20-1632.
[33] Chung, G. et al. (2013), “Antimicrobial stewardship”, Virulence, Vol. 4/2, pp. 151-157, https://doi.org/10.4161/viru.21626.
[72] CIDRAP (2019), “Poll: US public aware of antibiotic resistance but sketchy on details”, University of Minnesota Center for Infectious Disease Research and Policy, https://www.cidrap.umn.edu/news-perspective/2019/06/poll-us-public-aware-antibiotic-resistance-sketchy-details (accessed on 30 July 2020).
[99] Clements, A. et al. (2008), “Overcrowding and understaffing in modern health-care systems: Key determinants in meticillin-resistant Staphylococcus aureus transmission”, The Lancet Infectious Diseases, Vol. 8/7, pp. 427-434, https://doi.org/10.1016/s1473-3099(08)70151-8.
[82] CNISP/Public Health Agency of Canada (2020), “Healthcare-associated infections and antimicrobial resistance in Canadian acute care hospitals, 2014-2018”, Canada Communicable Disease Report, Vol. 46/5, pp. 99-112, https://doi.org/10.14745/ccdr.v46i05a01.
[87] Cowling, B. (ed.) (2012), “The Feedback Intervention Trial (FIT) — Improving hand-hygiene compliance in UK healthcare workers: A stepped wedge cluster randomised controlled trial”, PLoS ONE, Vol. 7/10, p. e41617, https://doi.org/10.1371/journal.pone.0041617.
[52] Cox, H. et al. (2015), “The need to accelerate access to new drugs for multidrug-resistant tuberculosis”, Bulletin of the World Health Organization, Vol. 93/7, pp. 491-497, https://doi.org/10.2471/blt.14.138925.
[169] DANMAP (2020), Use of Antimicrobial Agents and Occurrence of Antimicrobial Resistance in Bacteria from Food Animals, Food and Humans in Denmark, Danish Integrated Antimicrobial Resistance Monitoring and Research Programme, https://www.danmap.org/reports/2020 (accessed on 21 June 2020).
[25] Das, J. et al. (2016), “The impact of training informal health care providers in India: A randomized controlled trial”, Science, Vol. 354/6308, pp. aaf7384-aaf7384, https://doi.org/10.1126/science.aaf7384.
[14] Davey, P. et al. (2017), “Interventions to improve antibiotic prescribing practices for hospital inpatients”, Cochrane Database of Systematic Reviews, https://doi.org/10.1002/14651858.cd003543.pub4.
[113] de Figueiredo, A. et al. (2020), “Mapping global trends in vaccine confidence and investigating barriers to vaccine uptake: A large-scale retrospective temporal modelling study”, The Lancet, Vol. 396/10255, pp. 898-908, https://doi.org/10.1016/s0140-6736(20)31558-0.
[19] de Kraker, M. et al. (2017), “Good epidemiological practice: A narrative review of appropriate scientific methods to evaluate the impact of antimicrobial stewardship interventions”, Clinical Microbiology and Infection, Vol. 23/11, pp. 819-825, https://doi.org/10.1016/j.cmi.2017.05.019.
[110] Díaz Crescitelli, M. et al. (2020), “A meta-synthesis study of the key elements involved in childhood vaccine hesitancy”, Public Health, Vol. 180, pp. 38-45, https://doi.org/10.1016/j.puhe.2019.10.027.
[46] Dolk, F. et al. (2018), “Antibiotics in primary care in England: Which antibiotics are prescribed and for which conditions?”, Journal of Antimicrobial Chemotherapy, Vol. 73/suppl_2, pp. ii2-ii10, https://doi.org/10.1093/jac/dkx504.
[34] Donà, D. et al. (2020), “Implementation and impact of pediatric antimicrobial stewardship programs: A systematic scoping review”, Antimicrobial Resistance & Infection Control, Vol. 9/1, https://doi.org/10.1186/s13756-019-0659-3.
[10] dos Santos, R. et al. (2018), “Antimicrobial stewardship through telemedicine and its impact on multi-drug resistance”, Journal of Telemedicine and Telecare, Vol. 25/5, pp. 294-300, https://doi.org/10.1177/1357633x18767702.
[130] Dutil, L. et al. (2010), “Ceftiofur resistance in Salmonella enterica Serovar Heidelberg from chicken meat and humans, Canada”, Emerging Infectious Diseases, Vol. 16/1, pp. 48-54, https://doi.org/10.3201/eid1601.090729.
[59] EAHP (2019), 2019 EAHP Medicines Shortages Report: Medicines Shortages in European Hospitals, European Association of Hospital Pharmacists, Brussels, https://www.eahp.eu/practice-and-policy/medicines-shortages.
[195] EAWAG (2007), Annual Report 2007, Swiss Federal Institute of Aquatic Science and Technology, https://www.eawag.ch/fileadmin/Domain1/About/Portraet/Jahresbericht/Reports/eawag_annualreport_07.pdf.
[115] ECDC (2017), Catalogue of Interventions Addressing Vaccine Hesitancy, European Centre for Disease Prevention and Control, Stockholm, https://doi.org/10.2900/654210.
[186] EPR (2020), “Limiting antibiotic manufacturing discharge in Indian wastewater”, European Pharmaceutical Review, https://www.europeanpharmaceuticalreview.com/article/115074/limiting-antibiotic-manufacturing-discharge-in-indian-wastewater/ (accessed on 2 February 21).
[197] Eriksen, J. et al. (2017), “High adherence to the ‘Wise List’ treatment recommendations in Stockholm: A 15-year retrospective review of a multifaceted approach promoting rational use of medicines”, BMJ Open, Vol. 7/4, p. e014345, https://doi.org/10.1136/bmjopen-2016-014345.
[150] European Commission (2009), Directive 2009/128/EC of the European Parliament and of the Council of 21 October 2009 Establishing a Framework for Community Action to Achieve the Sustainable Use of Pesticide, European Commission, https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=celex%3A32009L0128.
[151] Eurostat (2022), Pesticide Sales, https://ec.europa.eu/eurostat/databrowser/view/AEI_FM_SALPEST09/bookmark/table?lang=en&bookmarkId=53792fd3-191d-4201-aab5-c01c67fd927c (accessed on 26 October 2022).
[154] FAO (2020), Antimicrobial Resistance (AMR) in Relation to Pesticide Use in Plant Production, Food and Agriculture Organization of the United Nations, http://www.fao.org/3/cb0660en/CB0660EN.pdf.
[133] FAO (2020), Antimicrobial Resistance: Key Facts, Food and Agriculture Organization of the United Nations, http://www.fao.org/antimicrobial-resistance/background/what-is-it/en/ (accessed on 22 January 2020).
[139] FAO (2020), Tackling Antimicrobial Use and Resistance in Dairy Cattle, Food and Agriculture Organization of the United Nations, https://doi.org/10.4060/cb2201en.
[175] FAO (2018), More People, More Food, Worse Water? A Global Review of Water Pollution from Agriculture, Water, Water, Land and Ecosystems (WLE) Program of the CGIAR, International Water Management Institute (IWMI), Food and Agriculture Organization, http://www.fao.org/policy-support/tools-and-publications/resources-details/en/c/1144303/.
[159] FAO (1997), Hazard Analysis and Critical Control Points (HACCP) Systems and Guidelines for its Application: Annex to CAC/RCP 1-1969, Rev. 3, Food and Agriculture Organization of the United Nations, https://www.fao.org/3/y1579e/y1579e03.htm.
[158] FAO (n.d.), Food Safety, Food and Agriculture Organization of the United Nations, http://www.fao.org/antimicrobial-resistance/key-sectors/food-safety/en/.
[134] FAO/DMOEF (2019), Tackling antimicrobial use and resistance in pig production: Lessons learned from Denmark, Food and Agriculture Organization of the United Nations, https://www.fao.org/documents/card/en?details=CA2899EN%2f.
[155] FAO/WHO (2019), Joint FAO/WHO Expert Meeting in Collaboration with OIE on Foodborne AntimicrobialResistance: Role of the Environment, Crops and Biocides – Meeting Report, Microbiological Risk Assessment Series no. 34, World Health Organization, https://apps.who.int/iris/handle/10665/332387.
[147] FAO/WOAH/WHO (2019), Monitoring Global Progress on Addressing Antimicrobial Resistance: Analysis Report of the Second Round of Results of AMR Country Self-Assessment Survey 2018, World Health Organization, https://apps.who.int/iris/handle/10665/273128.
[126] FDA (2020), Timeline of FDA Action on Antimicrobial Resistance, United States Food and Drug Administration, https://www.fda.gov/animal-veterinary/antimicrobial-resistance/timeline-fda-action-antimicrobial-resistance.
[105] Feldman, M. et al. (2019), “A promising bioconjugate vaccine against hypervirulentKlebsiella pneumoniae”, Proceedings of the National Academy of Sciences, Vol. 116/37, pp. 18655-18663, https://doi.org/10.1073/pnas.1907833116.
[185] Fick, J. et al. (2009), “Contamination of surface, ground, and drinking water from pharmaceutical production”, Environmental Toxicology and Chemistry, Vol. 28/12, p. 2522, https://doi.org/10.1897/09-073.1.
[62] Filippini, M., F. Heimsch and G. Masiero (2014), “Antibiotic consumption and the role of dispensing physicians”, Regional Science and Urban Economics, Vol. 49, pp. 242-251, https://doi.org/10.1016/j.regsciurbeco.2014.07.005.
[22] Fischer, M. et al. (2020), “Non-infection-related and non-visit-based antibiotic prescribing is common among Medicaid patients”, Health Affairs, Vol. 39/2, pp. 280-288, https://doi.org/10.1377/hlthaff.2019.00545.
[29] Fischer, M. et al. (2003), “Conversion from intravenous to oral medications”, Archives of Internal Medicine, Vol. 163/21, p. 2585, https://doi.org/10.1001/archinte.163.21.2585.
[66] Formoso, G. et al. (2013), “Feasibility and effectiveness of a low cost campaign on antibiotic prescribing in Italy: Community level, controlled, non-randomised trial”, BMJ, Vol. 347, p. f5391, https://doi.org/10.1136/bmj.f5391.
[177] Fouz, N. et al. (2020), “The contribution of wastewater to the transmission of antimicrobial resistance in the environment: Implications of mass gathering settings”, Tropical Medicine and Infectious Disease, Vol. 5/1, p. 33, https://doi.org/10.3390/tropicalmed5010033.
[36] Gerber, J. et al. (2013), “Effect of an outpatient antimicrobial stewardship intervention on broad-spectrum antibiotic prescribing by primary care pediatricians”, JAMA, Vol. 309/22, p. 2345, https://doi.org/10.1001/jama.2013.6287.
[94] Gerolemou, L. et al. (2014), “Simulation-based training for nurses in sterile techniques during central vein catheterization”, American Journal of Critical Care, Vol. 23/1, pp. 40-48, https://doi.org/10.4037/ajcc2014860.
[2] Getahun, H. et al. (2020), “Tackling antimicrobial resistance in the COVID-19 pandemic”, Bulletin of the World Health Organization, Vol. 98/7, pp. 442-442A, https://doi.org/10.2471/blt.20.268573.
[116] Gibson, D. et al. (2017), “Mobile phone-delivered reminders and incentives to improve childhood immunisation coverage and timeliness in Kenya (M-SIMU): A cluster randomised controlled trial”, The Lancet Global Health, Vol. 5/4, pp. e428-e438, https://doi.org/10.1016/s2214-109x(17)30072-4.
[40] Giry, M. et al. (2016), “Acceptability of antibiotic stewardship measures in primary care”, Médecine et Maladies Infectieuses, Vol. 46/6, pp. 276-284, https://doi.org/10.1016/j.medmal.2016.02.001.
[60] Goldacre, B. et al. (2019), “Do doctors in dispensing practices with a financial conflict of interest prescribe more expensive drugs? A cross-sectional analysis of English primary care prescribing data”, BMJ Open, Vol. 9/2, p. e026886, https://doi.org/10.1136/bmjopen-2018-026886.
[162] Government of Japan (2016), National Action Plan on Antimicrobial Resistance (2016-2020), https://www.mhlw.go.jp/file/06-Seisakujouhou-10900000-Kenkoukyoku/0000138942.pdf.
[168] Hammerum, A. et al. (2007), “Danish integrated antimicrobial resistance monitoring and research program”, Emerging Infectious Diseases, Vol. 13/11, pp. 1633-1639, https://doi.org/10.3201/eid1311.070421.
[97] Han, M. and X. Zhang (2020), “Impact of medical professionals on Carbapenem-resistant Pseudomonas aeruginosa: Moderating effect of workload based on the panel data in China”, BMC Health Services Research, Vol. 20/1, https://doi.org/10.1186/s12913-020-05535-5.
[199] HCWH-Europe (2013), Unused Pharmaceuticals Where Do They End Up? A Snapshot of European Collection Schemes, Health Care Without Harm Europe, https://noharm-europe.org/sites/default/files/documents-files/4646/2013-12%20Unused%20pharmaceuticals.pdf.
[104] Hegerle, N. et al. (2018), “Development of a broad spectrum glycoconjugate vaccine to prevent wound and disseminated infections with Klebsiella pneumoniae and Pseudomonas aeruginosa”, PLoS ONE, Vol. 13/9, p. e0203143, https://doi.org/10.1371/journal.pone.0203143.
[192] He, Y. et al. (2020), “Antibiotic resistance genes from livestock waste: Occurrence, dissemination, and treatment”, npj Clean Water, Vol. 3/1, https://doi.org/10.1038/s41545-020-0051-0.
[131] Hiki, M. et al. (2015), “Decreased resistance to broad-spectrum cephalosporin in Escherichia coli from healthy broilers at farms in Japan after voluntary withdrawal of ceftiofur”, Foodborne Pathogens and Disease, Vol. 12/7, pp. 639-643, https://doi.org/10.1089/fpd.2015.1960.
[194] Hocquet, D., A. Muller and X. Bertrand (2016), “What happens in hospitals does not stay in hospitals: Antibiotic-resistant bacteria in hospital wastewater systems”, Journal of Hospital Infection, Vol. 93/4, pp. 395-402, https://doi.org/10.1016/j.jhin.2016.01.010.
[120] Hoelzer, K. et al. (2017), “Antimicrobial drug use in food-producing animals and associated human health risks: What, and how strong, is the evidence?”, BMC Veterinary Research, Vol. 13/1, https://doi.org/10.1186/s12917-017-1131-3.
[38] Høgli, J. et al. (2016), “An audit and feedback intervention study increased adherence to antibiotic prescribing guidelines at a Norwegian hospital”, BMC Infectious Diseases, Vol. 16/1, https://doi.org/10.1186/s12879-016-1426-1.
[27] Holstiege, J., T. Mathes and D. Pieper (2014), “Effects of computer-aided clinical decision support systems in improving antibiotic prescribing by primary care providers: A systematic review”, Journal of the American Medical Informatics Association, Vol. 22/1, pp. 236-242, https://doi.org/10.1136/amiajnl-2014-002886.
[16] Honda, H. et al. (2017), “Antimicrobial stewardship in inpatient settings in the Asia Pacific region: A systematic review and meta-analysis”, Clinical Infectious Diseases, Vol. 64/suppl_2, pp. S119-S126, https://doi.org/10.1093/cid/cix017.
[95] Houghton, C. et al. (2020), “Barriers and facilitators to healthcare workers’ adherence with infection prevention and control (IPC) guidelines for respiratory infectious diseases: a rapid qualitative evidence synthesis”, Cochrane Database of Systematic Reviews, https://doi.org/10.1002/14651858.cd013582.
[73] Huttner, B. et al. (2019), “How to improve antibiotic awareness campaigns: Findings of a WHO global survey”, BMJ Global Health, Vol. 4/3, p. e001239, https://doi.org/10.1136/bmjgh-2018-001239.
[128] Hu, Y. and B. Cowling (2020), “Reducing antibiotic use in livestock, China”, Bulletin of the World Health Organization, Vol. 98/5, pp. 360-361, https://doi.org/10.2471/blt.19.243501.
[74] Isales, C. (ed.) (2018), “Antibiotic use, knowledge and health literacy among the general population in Berlin, Germany and its surrounding rural areas”, PLoS ONE, Vol. 13/2, p. e0193336, https://doi.org/10.1371/journal.pone.0193336.
[140] Isomura, R., M. Matsuda and K. Sugiura (2018), “An epidemiological analysis of the level of biosecurity and animal welfare on pig farms in Japan and their effect on the use of veterinary antimicrobials”, Journal of Veterinary Medical Science, Vol. 80/12, pp. 1853-1860, https://doi.org/10.1292/jvms.18-0287.
[102] IVAC (2019), VIEW-hub Report: Global Vaccine Introduction and Implementation, Johns Hopkins Bloomberg School of Public Health, Baltimore, http://www.jhsph.edu/ivac (accessed on 28 July 2020).
[135] Jensen, H. and D. Hayes (2014), “Impact of Denmark’s ban on antimicrobials for growth promotion”, Current Opinion in Microbiology, Vol. 19, pp. 30-36, https://doi.org/10.1016/j.mib.2014.05.020.
[132] Kaneene, J. et al. (2008), “Changes in tetracycline susceptibility of enteric bacteria following switching to nonmedicated milk replacer for dairy calves”, Journal of Clinical Microbiology, Vol. 46/6, pp. 1968-1977, https://doi.org/10.1128/jcm.00169-08.
[117] Karing, A. (2018), “Social signaling and childhood immunization: A field experiment in Sierra Leone”, Innovations for Poverty Action, https://www.poverty-action.org/publication/social-signaling-and-childhood-immunization-field-experiment-sierra-leone (accessed on 17 July 2020).
[171] Keen, P. and M. Montforts (eds.) (2011), Antimicrobial Resistance in the Environment, John Wiley & Sons, Inc., Hoboken, NJ, USA, https://doi.org/10.1002/9781118156247.
[65] Kim, H. and J. Ruger (2008), “Pharmaceutical reform In South Korea and the lessons it provides”, Health Affairs, Vol. 27/Suppl1, pp. w260-w269, https://doi.org/10.1377/hlthaff.27.4.w260.
[7] Kiss, P. et al. (2020), “The impact of the COVID-19 pandemic on the care and management of patients with acute cardiovascular disease: A systematic review”, European Heart Journal - Quality of Care and Clinical Outcomes, Vol. 7/1, pp. 18-27, https://doi.org/10.1093/ehjqcco/qcaa084.
[100] Klein, E. (ed.) (2020), “Private patient rooms and hospital-acquired methicillin-resistant Staphylococcus aureus: A hospital-level analysis of administrative data from the United States”, PLoS ONE, Vol. 15/7, p. e0235754, https://doi.org/10.1371/journal.pone.0235754.
[121] Klous, G. et al. (2016), “Human–livestock contacts and their relationship to transmission of zoonotic pathogens: A systematic review of literature”, One Health, Vol. 2, pp. 65-76, https://doi.org/10.1016/j.onehlt.2016.03.001.
[47] Kruk, M. (ed.) (2020), “Antibiotic prescription practices in primary care in low- and middle-income countries: A systematic review and meta-analysis”, PLoS Medicine, Vol. 17/6, p. e1003139, https://doi.org/10.1371/journal.pmed.1003139.
[89] Lacey, G. et al. (2020), “The impact of automatic video auditing with real-time feedback on the quality and quantity of handwash events in a hospital setting”, American Journal of Infection Control, Vol. 48/2, pp. 162-166, https://doi.org/10.1016/j.ajic.2019.06.015.
[67] Lambert, M., G. Masters and S. Brent (2007), “Can mass media campaigns change antimicrobial prescribing? A regional evaluation study”, Journal of Antimicrobial Chemotherapy, Vol. 59/3, pp. 537-543, https://doi.org/10.1093/jac/dkl511.
[4] Langford, B. et al. (2021), “Antibiotic prescribing in patients with COVID-19: Rapid review and meta-analysis”, Clinical Microbiology and Infection, https://doi.org/10.1016/j.cmi.2020.12.018.
[112] Larson, H. et al. (2018), State of Vaccine Confidence in the EU, European Commission, Luxembourg, https://doi.org/10.2875/241099.
[111] Larson, H. et al. (2014), “Understanding vaccine hesitancy around vaccines and vaccination from a global perspective: A systematic review of published literature, 2007-2012”, Vaccine, Vol. 32/19, pp. 2150-2159, https://doi.org/10.1016/j.vaccine.2014.01.081.
[173] Larsson, D. et al. (2018), “Critical knowledge gaps and research needs related to the environmental dimensions of antibiotic resistance”, Environment International, Vol. 117, pp. 132-138, https://doi.org/10.1016/j.envint.2018.04.041.
[183] Larsson, D., C. de Pedro and N. Paxeus (2007), “Effluent from drug manufactures contains extremely high levels of pharmaceuticals”, Journal of Hazardous Materials, Vol. 148/3, pp. 751-755, https://doi.org/10.1016/j.jhazmat.2007.07.008.
[63] Lim, D. et al. (2011), “Australian dispensing doctors’ prescribing: Quantitative and qualitative analysis”, Medical Journal of Australia, Vol. 195/4, pp. 172-175, https://doi.org/10.5694/j.1326-5377.2011.tb03272.x.
[88] Livorsi, D. et al. (2018), “Evaluation of barriers to audit-and-feedback programs that used direct observation of hand hygiene compliance”, JAMA Network Open, Vol. 1/6, p. e183344, https://doi.org/10.1001/jamanetworkopen.2018.3344.
[148] Lomazzi, M. et al. (2019), “Antimicrobial resistance – Moving forward?”, BMC Public Health, Vol. 19/1, https://doi.org/10.1186/s12889-019-7173-7.
[11] Lucien, M. et al. (2021), “Antibiotics and antimicrobial resistance in the COVID-19 era: Perspective from resource-limited settings”, International Journal of Infectious Diseases, Vol. 104, pp. 250-254, https://doi.org/10.1016/j.ijid.2020.12.087.
[142] Manyi-Loh, C. et al. (2018), “Antibiotic use in agriculture and its consequential resistance in environmental sources: Potential public health implications”, Molecules, Vol. 23/4, p. 795, https://doi.org/10.3390/molecules23040795.
[37] Meeker, D. et al. (2016), “Effect of behavioral interventions on inappropriate antibiotic prescribing among primary care practices”, JAMA, Vol. 315/6, p. 562, https://doi.org/10.1001/jama.2016.0275.
[161] MHLW (2016), Summary of the Final Report on the Implementation of Mandatory HACCP Program in Food Industry Adopted by the Ad Hoc Panel on International Standardization of Food Hygiene Control, Ministry of Health, Labour and Welfare, Japan, https://www.mhlw.go.jp/english/topics/foodsafety/consideration/dl/summary_of_the_final_report.pdf.
[51] Micallef, C. et al. (2017), “Secondary use of data from hospital electronic prescribing and pharmacy systems to support the quality and safety of antimicrobial use: A systematic review”, Journal of Antimicrobial Chemotherapy, Vol. 72/7, pp. 1880-1885, https://doi.org/10.1093/jac/dkx082.
[144] Midtlyng, P., K. Grave and T. Horsberg (2011), “What has been done to minimize the use of antibacterial and antiparasitic drugs in Norwegian aquaculture?”, https://doi.org/10.1111/j.1365-2109.2010.02726.x.
[76] Moreira, L. (2018), “Health literacy for people-centred care: Where do OECD countries stand?”, OECD Health Working Papers, No. 107, OECD Publishing, Paris, https://doi.org/10.1787/d8494d3a-en.
[160] MUFG (2016), “The imminent mandatory implementation of HACCP in Japan”, Quarterly Journal of Public Policy & Management, Vol. 4, https://www.murc.jp/english/report/quarterly_journal/qj1604_07/ (accessed on 21 January 2021).
[188] Nappier, S. et al. (2020), “Antibiotic resistance in recreational waters: State of the science”, International Journal of Environmental Research and Public Health, Vol. 17/21, p. 8034, https://doi.org/10.3390/ijerph17218034.
[17] Nathwani, D. et al. (2019), “Value of hospital antimicrobial stewardship programs [ASPs]: A systematic review”, Antimicrobial Resistance & Infection Control, Vol. 8/1, https://doi.org/10.1186/s13756-019-0471-0.
[35] Navathe, A. and E. Emanuel (2016), “Physician peer comparisons as a nonfinancial strategy to improve the value of care”, JAMA, Vol. 316/17, p. 1759, https://doi.org/10.1001/jama.2016.13739.
[43] NCAS/ACSQHC (2016), Antimicrobial Prescribing Practices in Australian Hospitals: Results of the 2015 Hospital National Antibiotic Prescribing Survey, National Centre for Antimicrobial Stewardship and Australian Commission on Safety and Quality in Health Care.
[80] Núñez-Núñez, M. et al. (2018), “The methodology of surveillance for antimicrobial resistance and healthcare-associated infections in Europe (SUSPIRE): A systematic review of publicly available information”, Clinical Microbiology and Infection, Vol. 24/2, pp. 105-109, https://doi.org/10.1016/j.cmi.2017.07.014.
[125] OECD (2019), Pharmaceutical Residues in Freshwater: Hazards and Policy Responses, OECD Studies on Water, OECD Publishing, Paris, https://doi.org/10.1787/c936f42d-en.
[109] OECD (2019), “Vaccinations”, in Health at a Glance 2019: OECD Indicators, OECD Publishing, Paris, https://doi.org/10.1787/2700bb99-en.
[1] OECD (2018), Stemming the Superbug Tide: Just a Few Dollars More, OECD Publishing, Paris, https://doi.org/10.1787/2074319x.
[64] Park, S. et al. (2005), “Antibiotic use following a Korean national policy to prohibit medication dispensing by physicians”, Health Policy and Planning, Vol. 20/5, pp. 302-309, https://doi.org/10.1093/heapol/czi033.
[200] Persson, M., E. Sabelström and B. Gunnarsson (2009), “Handling of unused prescription drugs - Knowledge, behaviour and attitude among Swedish people”, Environment International, Vol. 35/5, pp. 771-774, https://doi.org/10.1016/j.envint.2008.10.002.
[68] Perz, J. (2002), “Changes in antibiotic prescribing for children after a community-wide campaign”, JAMA, Vol. 287/23, p. 3103, https://doi.org/10.1001/jama.287.23.3103.
[42] Plachouras, D. et al. (2018), “Antimicrobial use in European acute care hospitals: Results from the second point prevalence survey (PPS) of healthcare-associated infections and antimicrobial use, 2016 to 2017”, Eurosurveillance, Vol. 23/46, https://doi.org/10.2807/1560-7917.es.23.46.1800393.
[141] Postma, M. et al. (2016), “Reducing antimicrobial usage in pig production without jeopardizing production parameters”, Zoonoses and Public Health, Vol. 64/1, pp. 63-74, https://doi.org/10.1111/zph.12283.
[149] Pretty, J. and Z. Bharucha (2015), “Integrated pest management for sustainable intensification of agriculture in Asia and Africa”, Insects, Vol. 6/1, pp. 152-182, https://doi.org/10.3390/insects6010152.
[71] Prigitano, A. et al. (2018), “Antibiotic resistance: Italian awareness survey 2016”, Journal of Infection and Public Health, Vol. 11/1, pp. 30-34, https://doi.org/10.1016/j.jiph.2017.02.010.
[54] Pulcini, C. et al. (2017), “Ensuring universal access to old antibiotics: A critical but neglected priority”, Clinical Microbiology and Infection, Vol. 23/9, pp. 590-592, https://doi.org/10.1016/j.cmi.2017.04.026.
[179] Raven, K. et al. (2019), “Genomic surveillance of Escherichia coli in municipal wastewater treatment plants as an indicator of clinically relevant pathogens and their resistance genes”, Microbial Genomics, Vol. 5/5, https://doi.org/10.1099/mgen.0.000267.
[3] Rawson, T. et al. (2020), “Bacterial and fungal co-infection in individuals with coronavirus: A rapid review to support COVID-19 antimicrobial prescribing”, Clinical Infectious Diseases, Vol. 30/May, p. 28, https://doi.org/10.1093/cid.
[45] Ray, M. et al. (2019), “Antibiotic prescribing without documented indication in ambulatory care clinics: National cross sectional study”, BMJ, p. l6461, https://doi.org/10.1136/bmj.l6461.
[196] Regional Drug Expert Consortium (2011), “The ‘Wise List’- A comprehensive concept to select, communicate and achieve adherence to recommendations of essential drugs in ambulatory care in Stockholm”, Basic & Clinical Pharmacology & Toxicology, Vol. 108/4, pp. 224-233, https://doi.org/10.1111/j.1742-7843.2011.00682.x.
[119] Review on Antimicrobial Resistance (2015), Antimicrobials in Agriculture and the Environment: Reducing Unnecessary Use and Waste, Chaired by Jim O’Neill, Wellcome Trust.
[9] Riera, R. et al. (2021), “Delays and disruptions in cancer health care due to COVID-19 pandemic: Systematic review”, JCO Global Oncology 7, pp. 311-323, https://doi.org/10.1200/go.20.00639.
[184] Rodriguez-Valera, F. (ed.) (2011), “Pyrosequencing of antibiotic-contaminated river sediments reveals high levels of resistance and gene transfer elements”, PLoS ONE, Vol. 6/2, p. e17038, https://doi.org/10.1371/journal.pone.0017038.
[143] Rojo-Gimeno, C. et al. (2016), “Farm-economic analysis of reducing antimicrobial use whilst adopting improved management strategies on farrow-to-finish pig farms”, Preventive Veterinary Medicine, Vol. 129, pp. 74-87, https://doi.org/10.1016/j.prevetmed.2016.05.001.
[49] Roumeliotis, N. et al. (2019), “Effect of electronic prescribing strategies on medication error and harm in hospital: A systematic review and meta-analysis”, Journal of General Internal Medicine, Vol. 34/10, pp. 2210-2223, https://doi.org/10.1007/s11606-019-05236-8.
[163] Roy, A. et al. (2016), “Use of minimal-text posters to improve the microbial status of leafy greens and food contact surfaces in foodservice sites serving older adults”, Food Protection Trends, International Association for Food Protection.
[122] Ryan, M. (2019), “Evaluating the economic benefits and costs of antimicrobial use in food-producing animals”, OECD Food, Agriculture and Fisheries Papers, No. 132, OECD Publishing, Paris, https://doi.org/10.1787/f859f644-en.
[5] Saleem, Z. et al. (2019), “Point prevalence surveys of health-care-associated infections: A systematic review”, Pathogens and Global Health, Vol. 113/4, pp. 191-205, https://doi.org/10.1080/20477724.2019.1632070.
[18] Schweitzer, V. et al. (2019), “The quality of studies evaluating antimicrobial stewardship interventions: A systematic review”, Clinical Microbiology and Infection, Vol. 25/5, pp. 555-561, https://doi.org/10.1016/j.cmi.2018.11.002.
[187] Scott, T. et al. (2018), “Pharmaceutical manufacturing facility discharges can substantially increase the pharmaceutical load to U.S. wastewaters”, Science of The Total Environment, Vol. 636, pp. 69-79, https://doi.org/10.1016/j.scitotenv.2018.04.160.
[8] Scquizzato, T. et al. (2020), “Effects of COVID-19 pandemic on out-of-hospital cardiac arrests: A systematic review”, Resuscitation, Vol. 157, pp. 241-247, https://doi.org/10.1016/j.resuscitation.2020.10.020.
[178] Shekhawat, S., N. Kulshreshtha and A. Gupta (2020), “Tertiary treatment technologies for removal of antibiotics and antibiotic resistance genes from wastewater”, in Removal of Toxic Pollutants Through Microbiological and Tertiary Treatment, Elsevier, https://doi.org/10.1016/b978-0-12-821014-7.00001-0.
[24] Shively, N. et al. (2019), “Impact of a telehealth-based antimicrobial stewardship program in a community hospital health system”, Clinical Infectious Diseases, Vol. 71/3, pp. 539-545, https://doi.org/10.1093/cid/ciz878.
[41] Sikkens, J. et al. (2017), “Behavioral approach to appropriate antimicrobial prescribing in hospitals”, JAMA Internal Medicine, Vol. 177/8, p. 1130, https://doi.org/10.1001/jamainternmed.2017.0946.
[182] Singer, A. (ed.) (2017), “A comparison of extended spectrum β-lactamase producing Escherichia coli from clinical, recreational water and wastewater samples associated in time and location”, Plos ONE, Vol. 12/10, p. e0186576, https://doi.org/10.1371/journal.pone.0186576.
[157] Smith, J. and P. Fratamico (2016), “Escherichia coli and other Enterobacteriaceae: Food poisoning and health effects”, in Encyclopedia of Food and Health, Elsevier, https://doi.org/10.1016/b978-0-12-384947-2.00260-9.
[164] Soares, K. et al. (2013), “Evaluation of food safety training on hygienic conditions in food establishments”, Food Control, Vol. 34/2, pp. 613-618, https://doi.org/10.1016/j.foodcont.2013.06.006.
[75] Sørensen, K. et al. (2015), “Health literacy in Europe: Comparative results of the European health literacy survey (HLS-EU)”, The European Journal of Public Health, Vol. 25/6, pp. 1053-1058, https://doi.org/10.1093/eurpub/ckv043.
[78] Storr, J. et al. (2017), “Core components for effective infection prevention and control programmes: New WHO evidence-based recommendations”, Antimicrobial Resistance & Infection Control, Vol. 6/1, https://doi.org/10.1186/s13756-016-0149-9.
[83] Streefkerk, H. et al. (2020), “Electronically assisted surveillance systems of healthcare-associated infections: A systematic review”, Eurosurveillance, Vol. 25/2, https://doi.org/10.2807/1560-7917.es.2020.25.2.1900321.
[181] Su, J. et al. (2017), “Metagenomics of urban sewage identifies an extensively shared antibiotic resistome in China”, Microbiome, Vol. 5/1, https://doi.org/10.1186/s40168-017-0298-y.
[123] Tang, K. et al. (2019), “Comparison of different approaches to antibiotic restriction in food-producing animals: Stratified results from a systematic review and meta-analysis”, BMJ Global Health, Vol. 4/4, p. e001710, https://doi.org/10.1136/bmjgh-2019-001710.
[21] TATFAR (2015), Modified Delphi Process for Common Structure and Process Indicators for Hospital Antimicrobial Stewardship Programs, Transatlantic Taskforce on Antimicrobial Resistance, https://www.cdc.gov/drugresistance/pdf/summary_of_tatfar_recommendation_1.pdf.
[153] Taylor, P. and R. Reeder (2020), “Antibiotic use on crops in low and middle-income countries based on recommendations made by agricultural advisors”, CABI Agriculture and Bioscience, Vol. 1/1, https://doi.org/10.1186/s43170-020-00001-y.
[96] Teixeira Rodrigues, A. et al. (2016), “Determinants of physician antibiotic prescribing behavior: A 3 year cohort study in Portugal”, Current Medical Research and Opinion, Vol. 32/5, pp. 949-957, https://doi.org/10.1185/03007995.2016.1154520.
[190] Tell, J. et al. (2019), “Science‐based targets for antibiotics in receiving waters from pharmaceutical manufacturing operations”, Integrated Environmental Assessment and Management, Vol. 15/3, pp. 312-319, https://doi.org/10.1002/ieam.4141.
[145] Thaker, M. (2006), “Comparison of the effects of oral vaccination and different dietary antibiotic prophylactic treatment against Lawsonia intracellularis associated losses in a fattening pig production unit with high prevalence of porcine proliferative enteropathy (PPE)”, Tierärztl Umsch, Vol. 61, pp. 372-376.
[28] Thursky, K. et al. (2006), “Reduction of broad-spectrum antibiotic use with computerized decision support in an intensive care unit”, International Journal for Quality in Health Care, Vol. 18/3, pp. 224-231, https://doi.org/10.1093/intqhc/mzi095.
[198] Tong, A., B. Peake and R. Braund (2011), “Disposal practices for unused medications around the world”, Environment International, Vol. 37/1, pp. 292-298, https://doi.org/10.1016/j.envint.2010.10.002.
[61] Trottmann, M. et al. (2016), “Physician drug dispensing in Switzerland: Association on health care expenditures and utilization”, BMC Health Services Research, Vol. 16/1, https://doi.org/10.1186/s12913-016-1470-y.
[31] Tuon, F. et al. (2017), “Mobile health application to assist doctors in antibiotic prescription – An approach for antibiotic stewardship”, The Brazilian Journal of Infectious Diseases, Vol. 21/6, pp. 660-664, https://doi.org/10.1016/j.bjid.2017.08.002.
[124] Umwelt Bundesamt (2018), Umweltaspekte bei Verabreichung von Tierarzneimitteln, https://www.umweltbundesamt.de/umweltaspekte-bei-verabreichung-von#Landwirtschaft (accessed on 2021 January 16).
[174] UNEP (2017), Frontiers 2017: Emerging Issues of Environmental Concern, United Nations Environment Programme.
[15] Van Dijck, C., E. Vlieghe and J. Cox (2018), “Antibiotic stewardship interventions in hospitals in low-and middle-income countries: A systematic review”, Bulletin of the World Health Organization, Vol. 96/4, pp. 266-280, https://doi.org/10.2471/blt.17.203448.
[84] van Mourik, M. et al. (2017), “Designing surveillance of healthcare-associated infections in the era of automation and reporting mandates”, Clinical Infectious Diseases, Vol. 66/6, pp. 970-976, https://doi.org/10.1093/cid/cix835.
[156] von Seidlein, L. (ed.) (2015), “World Health Organization Global estimates and regional comparisons of the burden of foodborne disease in 2010”, Plos Medicine, Vol. 12/12, p. e1001923, https://doi.org/10.1371/journal.pmed.1001923.
[92] Wang, J. et al. (2019), “Implementation of infection prevention and control in acute care hospitals in Mainland China - A systematic review”, Antimicrobial Resistance & Infection Control, Vol. 8/1, https://doi.org/10.1186/s13756-019-0481-y.
[85] Weber, D. et al. (2012), “Completeness of surveillance data reported by the National Healthcare Safety Network: An analysis of healthcare-associated infections ascertained in a tertiary care hospital, 2010”, Infection Control & Hospital Epidemiology, Vol. 33/1, pp. 94-96, https://doi.org/10.1086/663344.
[23] Webster, P. (2020), “Virtual health care in the era of COVID-19”, The Lancet, Vol. 395/10231, pp. 1180-1181, https://doi.org/10.1016/s0140-6736(20)30818-7.
[58] WHO (2021), 2021 AWaRe classification, World Health Organization, https://apps.who.int/iris/handle/10665/345555.
[189] WHO (2020), “Annex 6: Points to consider for manufacturers and inspectors:environmental aspects of manufacturing for the prevention of antimicrobial resistance”, in WHO Expert Committee on Specifications for Pharmaceutical Preparations, WHO Technical Report Series, no. 1025, World Health Organization, https://apps.who.int/iris/handle/10665/331814.
[20] WHO (2019), Antimicrobial Stewardship Programmes in Health-care Facilities in Low- and Middle-income Countries: A WHO Practical Toolkit, World Health Organization, https://apps.who.int/iris/handle/10665/329404.
[127] WHO (2019), Highest Priority Critically Important Antimicrobials, World Health Organization, https://www.who.int/foodsafety/cia/en/ (accessed on 26 February 2021).
[103] WHO (2019), Pneumococcal Conjugate (PCV3) - Immunization Coverage Estimates by WHO Region, World Health Organization, Geneva.
[79] WHO (2018), Improving Infection Prevention and Control at the Health Facility Interim practical manual supporting implementation of the WHO Guidelines on Core Components of Infection Prevention and Control Programmes, World Health Organization, Geneva, https://apps.who.int/iris/handle/10665/279788.
[53] WHO (2018), Meeting Report Antibiotic Shortages: Magnitude, Causes and Possible Solutions, Norwegian Directorate of Health, Oslo, Norway, https://apps.who.int/iris/handle/10665/311288.
[193] WHO (2017), Safe Management of Wastes from Health-care Activities: A Summary, World Health Organization, https://apps.who.int/iris/handle/10665/259491.
[56] WHO (2017), WHO best-practice statement on the off-label use of bedaquiline and delamanid for the treatment of multidrug-resistant tuberculosis, World Health Organization, https://apps.who.int/iris/handle/10665/258941.
[118] WHO (2017), WHO Guidelines on Use of Medically Important Antimicrobials in Food-producing Animals, World Health Organization, https://apps.who.int/iris/handle/10665/258970.
[77] WHO (2016), Guidelines on core components of infection prevention and control programmes at the national and acute health care facility level, World Health Organization, Geneva, https://apps.who.int/iris/handle/10665/251730.
[69] WHO (2015), Antibiotic Resistance: A Multi-Country Public Awareness Survey, World Health Organization, https://apps.who.int/iris/handle/10665/194460.
[57] WHO (2013), The Use of Bedaquiline in the Treatment of Multidrug-resistant Tuberculosis, World Health Organization, https://apps.who.int/iris/handle/10665/84879.
[101] WHO (2012), Global Vaccine Action Plan 2011-2020, World Health Organization, Geneva, https://apps.who.int/iris/handle/10665/78141.
[136] WHO (2002), Impact of Antimicrobial Growth Promoter Termination in Denmark: The WHO International Review Panel’s Evaluation of the Termination of the Use of Antimicrobial Growth Promoters in Denmark, World Health Organization, https://apps.who.int/iris/handle/10665/68357.
[55] WHO (n.d.), Frequently Asked Questions on Bedaquiline, World Health Organization.
[114] WHO/Europe (2017), Best Practice Guidance: How to Respond to Vocal Vaccine Deniers in Public, WHO Regional Office for Europe, https://apps.who.int/iris/handle/10665/343301.
[152] WHO/FAO/WOAH (2022), Tripartite AMR Country Self-Assessment Survey (TrACSS) 2021-2022, World Health Organization, https://amrcountryprogress.org/#/map-view (accessed on 23 March 2022).
[176] WHO/WOAH/FAO (2018), Technical Brief on Water, Sanitation, Hygiene and Wastewater Management to Prevent Infections and Reduce the Spread of Antimicrobial Resistance, World Health Organization, https://apps.who.int/iris/handle/10665/332243.
[167] Williams, M. et al. (2020), “Changes in salmonella contamination in meat and poultry since the introduction of the pathogen reduction and hazard analysis and critical control point rule”, Journal of Food Protection, Vol. 83/10, pp. 1707-1717, https://doi.org/10.4315/jfp-20-126.
[129] WOAH (2020), Annual Report on Antimicrobial Agents Intended for Use in Animals: Better Understanding of the Global Situation, World Organisation for Animal Health, https://www.woah.org/app/uploads/2022/06/a-sixth-annual-report-amu-final.pdf.
[166] Wu, X. et al. (2019), “Evaluation of the containment of antimicrobial-resistant salmonella species from a hazard analysis and critical control point (HACCP) and a non-HACCP pig slaughterhouses in Northeast Thailand”, Pathogens, Vol. 9/1, p. 20, https://doi.org/10.3390/pathogens9010020.
[6] Yap, F. et al. (2004), “Increase in methicillin-resistant Staphylococcus aureus acquisition rate and change in pathogen pattern associated with an outbreak of severe acute respiratory syndrome”, Clinical Infectious Diseases, Vol. 39/4, pp. 511-516, https://doi.org/10.1086/422641.
[39] Zetts, R. et al. (2020), “Primary care physicians’ attitudes and perceptions towards antibiotic resistance and outpatient antibiotic stewardship in the USA: A qualitative study”, BMJ Open, Vol. 10/7, p. e034983, https://doi.org/10.1136/bmjopen-2019-034983.
[50] Zhao, H. et al. (2021), “Appropriateness of antibiotic prescriptions in ambulatory care in China: A nationwide descriptive database study”, The Lancet Infectious Diseases, https://doi.org/10.1016/s1473-3099(20)30596-x.
[91] Zingg, W. et al. (2015), “Hospital organisation, management, and structure for prevention of health-care-associated infection: A systematic review and expert consensus”, The Lancet Infectious Diseases, Vol. 15/2, pp. 212-224, https://doi.org/10.1016/s1473-3099(14)70854-0.